Introduction
I look at its value as a colourant, its anti-oxidative properties and as an antimicrobial. After all is said and done I still am extremely sceptical of the claims of suppliers who maintain that their brines contain no nitrogen – only plant extracts. I discussed the matter at length with several authorities over the world for many years and we are in agreement on our scepticism. In personal correspondence with CHR Hansen on the matter, they responded as follows, saying, “Regarding alternative curing brines based on plant extract or other alternatives to NO2- or NO3- then we have been monitoring this area for some time – and I can surely appreciate your sound scepticism towards some solutions claiming no hidden nitrite/nitrates as seen in product from Southern Europe. We have conducted internal trials and also searched the web for other studies and it is becoming clear that the colour formation is based on a quite low amount of in-going nitrite, which is sufficient to provide the desired colour (at the recommended dosage of 1%), while at such relative low in-going nitrite level it is not possible to detect any residual nitrite with some analytical methods.”
There is, however, sound footing in the literature, that point to enhanced colour development in synergistic plant matter- nitrate/ nitrite systems. When plant-based fermentation brines came about I was personally very sceptical, but after I discovered historical precedence as well as the tremendous anti-oxidative value it has mainly based on the work of Aloys L. Tappel, I completely changed my view on it, now viewing it as a highly productive method.
I set out to design a curing system using beetroot and micro particle technology and sought ways to balance the nitrate-ascorbate content so that a brine can be designed where neither of the two is added from synthesised sources.
I finally provide the processing steps for such a brine using the starter culture of CHR Hansen which I think is the most developed in the world.
Towards Beet in a Curing Brine
-> Beetroot Analysed

Kale (2018)
The protein content is very small and I wonder what the source of the nitrogen is. Protein was calculated as % Protein = Nitrogen × 6.25. Nitrogen is found in at least three sources in beetroot being (1) as part of the colour fractions, (2) as nitrate and (3) a very small percentage as actual protein. We cannot only do the proximate analysis, we have to specifically test for nitrates. The amount of nitrates in the leaves and the tubers of the beetroot varies greatly which is something you will see throughout our study. There are ways that a “more constant” percentage can be secured. It is known that factors such as nitrogen content in fertilisers and environmental factors such as stress play a role in the final percentage nitrate in beetroot and limiting the procurement from one farm, for example, will contribute to this.
Salehzadeh (2020) reports that “the nitrate concentration in vegetables is determined via different factors, such as the biological properties of the plant, light intensity, soil type, temperature, moisture, plant density/seed, plant maturity, growth period, harvest time, plant size, storage time and nitrogen source [Tamme (2006), Hmelak (2013)]. In addition, we found a significant relationship between vegetable nitrate levels and vegetable categories, vegetable type and sampling season (p < 0.05). Based on studies by Pourmoghim et al., Tabatabaee et al. and Ierna, nitrate levels were higher in leafy, root and Fruit vegetables, respectively [Pretty (2000), Bednar (1994), Williams (2016)]. Moreover, the nitrate levels in vegetables in the fall were higher than that in the spring.” (Salehzadeh, 2020)
“This difference can be attributed to various factors, such as the duration and intensity of light radiation, soil and weather temperature, moisture and plant age [Pavlou (2007), Pourmoghim (2010), RAHMANI (2006), Ramachandran (2005)]. Extreme temperatures reduce the nitrate levels of plants through the assimilation process. Since there are more cloudy days in the fall than in the spring and the air temperature is lower, this results in lower nitrate assimilation during the fall, so the vegetables have higher nitrate levels [Boroujerdnia (2007), Shahlaei (2007)].” (Salehzadeh, 2020)
The proximate analysis from DCD’ed beetroot will be different from samples from non-DCD’ed beetroot. The process invented by Green Cell Technologies has the surprising result of “unlocking” greater protein values in other studies they have done. It will therefore be interesting to repeat the Kale (2018) analysis after the complete beetroot has been processed using DCD Technology.
I suggest that samples of beetroot processed by Green Cell Technologies, be sent to SGS and that the nitrate content be determined separately from the proximate analysis.
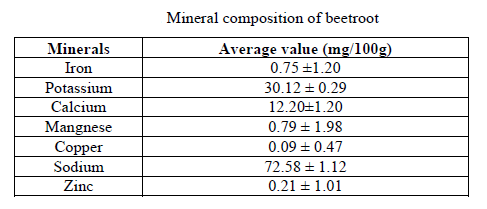
Kale (2018)

Kale (2018)
These results are further reasons for my proposal to test for these results following DCD Treatment with the equipment of Green Cell Technologies. Total sugars are 7.93 of which reducing sugars is 4.20% which is over 50% of the total sugar in the juice. Sucrose, which is by far the biggest component of sugar in beetroot is a non-reducing sugar while glucose and fructose, both reducing sugars are only a tiny fraction of the overall sugar content. This means that after the juice extraction process, most of the sugar is still in dry matter. If beetroot juice is used (the type used by Kale), the nutritive value of having more sucrose in the juice than fructose and glucose will be lost. In chapter 3 of the historical review, I indicate the value of using beetroot sugar in bacon production due to an improved caramelization during frying resulting in “crispier” bacon compared to using reducing sugars.
In light of these results and the problem I have with the determination of the protein content, I have contacted the owners of Green Cell Technologies and requested if the Kale (2018) analysis can be repeated on whole beetroots processed by them.
-> Designing a Beet-Based Brine
In the proposed method for processing the beetroot, I would want to experiment using the complete plant and not just the tuber with the equipment of Green Cell Technologies.
Ceclu (2020) gives nutritionally important information about both the tuber and the leaves in the following table.
He does not give the nitrate levels. For this I turn to an EFA report, The EFSA Journal (2008) 689, 1-79, Nitrate in vegetables Scientific Opinion of the Panel on Contaminants in the Food chain, which I attach in the reference section. They list beet under the heading leafy vegetables and separately under “Roots and Tubers”. I am assuming the one is the leave only and the other the tuber only. The median nitrate for the leafy part is 1770 mg/kg and 1100mg/kg for the beetroot under their analysis of “Roots and Tubers.”
Salehzadeh (2020) mg/kg nitrate level from different studies as 93.4 [Tamme (2006)], 1026 [EFS (2008)] 1250 [Bill], 1021 and from Sanandaj farms in Iran, 439 mg/kg.
This means that if we add 200mL to 1kg meat to the product processes with the Dynamic Cell Disruption (DCD) technology of Green Cell Technologies, we have the following. I assume a weight loss during smoking of 8% which will then yield a nett of 1.1kg and I further assume that only water is lost in the process. This will mean that we will add the following to the meat:
-> If we use the tubers: 1100mg/kg or 1.1mg per g. We add 200mL (assuming 1g = 1mL) This is then 220mg in the 200mL added which is added to 1.1kg. Using our assumption of 1kg being equal to 1L, we then know that we added 200mg/Kg (assuming we lost only water during heat treatment which is the same as adding 200ppm to bacon.
-> If we use the leafy part, ad using the same logic, we will add 354mg in 200mL to 1.1kg meat which is 321mG/Kg or 321ppm in the final product.
-> we know that vitamin C in the leave is 7.2mg/ 100mL in the tuber and 30 in the leave. It translates to adding 14.4mg and 60mg to the bacon when we used the tuber and the leaves respectively which is 13.09 and 54.54 ppm or mg/kg final product.
I am designing a hypothetical brine and would handle it as follows. Following Ceclu’s table and values, we know that both the tuber and the leaves have 91% water. I want to increase the vitamin C content and reduce the nitrites. How do we do that?
- Drying some of the mixes;
- Reduce the nitrate content in some of the mix;
- Combining it to the right overall result
- Extraction
->> Drying
We first look at drying it. We can either reduce it to powder and re-hydrate it for use or we can de-hydrate it to the right concentration. I lean towards the second option due to the sensitivity of betalains to reduced moisture but practical difficulties in distributing and storing the raw material will ultimately decide. I can also think of several ways that the partially dehydrated product can be stored for extended periods of time and production can be done at a site close to where it will be used.
I want to use the bulb and the leaves together and I will dry the products following DCD treatment and partially hydrate them or simply partially dry them. Dehydration and hydration have been discussed by Székely (2016). I give the full article below in the reference section. If you are interested in looking at it, in their colour evaluation they omit a description for the parameters, but these are customarily L* for luminosity, b* for yellow colour intensity and a* for red colour intensity.
In their study, they investigated two types of drying, one under vacuum and the other in an oven. They found that vacuum drying is a more favourable method to preserve the nutritional characteristics, but, as I noted above, it is necessary to dry the samples for the same moisture content and water activity because high moisture content and water activity affect the extraction and stability of betalain. The important point is that whichever method is used, work will have to be done to understand the effect of the various nutritional factors and colour and not just the effect of drying on nitrate content.
->> Cooking
The way that I would propose reducing the nitrite concentration I would propose is either cooking the beetroot or increasing the water content. The concern would be to “dilute” the value of the other properties of the beetroot. On the other hand, cooking it may likewise reduce the other nutritional factors which must be tested and validated.
The greatest clarity related to cooking and nitrate content comes to us from the study of Salehzadeh (2020). This landmark study not only focuses on beetroot but on the broad category of vegetables. In looking at beet as part of the broader category of vegetables, they focused on the beet leaves.
They conducted a study to investigate the effect of home cooking processes on nitrate reduction which they correlated to an assessment of health risks of nitrate in vegetables for consumers. They concluded that “the obtained research results indicate that “nitrate concentration in leafy vegetables was higher than root and fruit vegetables and these values were higher in autumn than in spring. The results of this study indicate that the cooking process reduces vegetable nitrate levels and lowers the health risk of eating raw vegetables, while the frying process increases the nitrate level in vegetables and thus increases their health risk.” (Salehzadeh, 2020)
Analysing dried beetroot, the minimum level was reduced by 18% from 3120 mg/kg to 2541 after cooking and the maximum levels analysed were reduced by 13% from 15394 to 13393 mg/kg. Fresh beetroot analysed saw a reduction of nitrate from 439 to 410 or 6.6%.
The beetroot was cooked, using a stainless steel pot, filled with distilled water (400 ml), and the water in the pot was raised to 90°C. After 15 minutes, the cooked vegetables were filtered and ground into powder after dehydrating and drying.
They state that “cooking changing the amount of nitrates in vegetables has also been reported by other researchers. Prasad et al. reported that the cooking process reduced the nitrate content of vegetables by 47 to 56% [Prasad (2008)]. Sadeghi et al. showed that the cooking process caused a slight increase in nitrate levels of some vegetable categories and a slight decrease in nitrate levels in others [Sadeghi (2013)].”
How this happened is interesting since it is similar to the study we looked at in a previous feedback report, Project Feedback May 2022, the paragraph entitles “Trial Set 3: A Catholyte Wash Step” where the work of Franco–Avila et al (2014) is sited. The basic method of nitrate reduction described by Franco-Avila (2014) is the same as that investigated by Salehzadeh (2020) except the latter includes a cooking step and the total time spent in the water is much longer than done in the former study. Salehzadeh (2020) speculate that “nitrates have a high tendency to dissolve in water, and when vegetables are immersed in water, the nitrates tend to move along the diffusion gradient from being more concentrated (inside the vegetable) to being less concentrated (inside the water in which the vegetable is located). Increasing temperature and time will facilitate the diffusion process and the movement of nitrates from the inside the vegetable into the distilled water, and finally, more nitrates will be released from the vegetable into the water, which will reduce the nitrate levels in the vegetable [Prasad (2008), Huarte-Mendicoa (1997)].” (Salehzadeh, 2020)
The other way that nitrites itself is reduced is of course through heating the meat as happens in the heating step of bacon and ham processing. We will investigate this when we get to smoking and heat treatment in our historical review. Related to cooking as a way of reducing nitrites, on the other hand, Karoline (2020) references Merino et al. who “did not find any effect of boiling on the nitrite content in pork and beef sausage; in contrast, frying reduced nitrite content by about 50%.” Besides this, they also say that “twenty‐four hours after the addition of nitrite, the authors observed a decrease in the amount of nitrite in all meat products tested (i.e., liver pate, pork/beef sausage, lunch chicken sausage and grilled sausage).” (Karolina, 2020)
Salehzadeh (2020) also examined the effect of home frying at 190oC. They explain this by saying that “Studies have shown that increasing the concentration of vegetable nitrates after frying can be related to the reduction of the vegetable’s mass, which ultimately results in the condensation of nitrates in the sample and the amount of oil consumed. This can be explained by the fact that during the frying process, vegetable nitrates remain at the same level, but the volume or weight of the vegetable is reduced; this causes the fixed amount of nitrates to be crowded into a lower volume.” (Salehzadeh, 2020)
In their discussion on frying, they point to an unexpected source for nitrite which I have not previously considered during tests for nitrite content after frying. They explain it thus: “The type of oil used for frying may be effective in reducing the number of nitrates in the vegetable; that is, the oil itself may contain nitrates. This hypothesis has been well documented in that the roots of immature plants, such as soybeans, have specific bacteria that stabilize elemental nitrogen to ammonia in plants. The ammonia is converted into nitrates through the nitrification process and by other microorganisms in soy plants, and because soybean oil was used in this study, the oil itself could contain nitrates and may have contaminated the fried vegetables [Prasad (2008), Hill (1996)].” (Salehzadeh, 2020)
->> Mixing
Using the techniques discussed above, it is conceivable that a system can be designed to produce a beetroot mix and processing parameters that would effectively “interfere” with the nitrate and vitamin C content and if this can be done in a way to satisfy the requirements for the Agency for Food Safty and the rules related to processing and extraction will have to be carefully considered. The technology of Green Cell Technologies presents very exciting opportunities by reducing the beetroot (the complete plant) to microparticles.
Under mixing, we must also leave the door open to use a mix between different plant-based materials. Prof Tappel whom I mentioned in the opening summary found that plant matter has the greatest impact on human health when used in combinations. This is something almost completely lacking from the investigations we will look at in a moment – the value of plant matter for colour development, as antioxidant and its antimicrobial value. There is work, which I did not get to this month, which shows the synergistic value and certain plants become counterproductive when used in combination. This is a field that must be approached with the greatest care and focus.
-> Extraction
The final option is of course extraction. That certain elements of plants contribute to colour development is certain. Karoline (2020) provides a roadmap through the legislative framework as far as the EU is concerned. Why is this important? It gives us an understanding of the process that European ingredient suppliers had to go through to have their products approved. Similar processes apply in the UK. Karoline (2020) describes the requirements and considerations as follows. “The application of new food additives, flavourings and enzymes is regulated by Regulation (EC) 1331/2008 of the European Parliament and of the Council. After receipt of an application, the Antioxidants 2020, 9, 711 8 of 31 European Commission initiates a procedure. The Commission also requests the opinion of the European Food Safety Authority (EFSA). More detailed information about applications is regulated by Commission Regulation (EU) 234/2011. This regulation clearly indicates that use of food enzymes and additives should be technologically justified. According to 234/2011, applications must be drafted according to the model presented in the Annex. In general, applications must contain: a letter, a technical dossier (all administrative data, and all data requested for the risk assessment and risk management) and a summary of the dossier. Toxicological tests must be performed according to Directive 2004/10EC (if tests are performed in EU) or the OECD Principles of Good Laboratory Practice (GLP) (OECD, 1998) (outside the EU). In a detailed manner, Commission Regulation 234/2011 specifies the kind of information that should be presented in the applications; for this reason, the issue will not be widely discussed, and only some issues will be mentioned. Information that must be included in the application includes the identity of the food additive; its stability, reaction and impact in the foods in which it is used; and biological and toxicological data. The Regulations also specify that specific, separate data is required for risk assessment of food enzymes, additives and flavorings. On the official website of the European Union, the practical guide for the authorization procedure can be found. Furthermore, point A.09 of the Summary Report of the Standing Committee on Plants, Animals, Food and Feed held in Brussels on 17 September 2018 contains information related to plant extracts. Depending on their purpose (substance of flavoring properties or substance of technological function) their status can be considered as flavoring or food additive (in which case, food additive legislation shall apply). To provide more clarity, in 2019 EFSA created a “Best Practice Guidance Document” concerning plant extracts and their legislation status. The guide was created to eliminate all ambiguities related to the legal status of extracts resulting from their traditional use as a flavoring agent. According to the guide, to determine the correct regulatory status of plant extracts, the intended application in the final food and extraction process should be considered.” (Karoline, 2020)
“In the case of extracts, Directive 2009/32/EC states that extraction solvents cannot contain toxicologically dangerous substances and should satisfy the specific criteria of purity. Moreover, the Directive also lists extraction solvents which are permitted for use in extraction processes (ethanol, acetone, butyl acetate, etc.) and solvents for which some specific conditions should be applied (hexane, dichloromethane, etc.). Depending on the target substances to be extracted (polyphenols, anthocyanins), it is possible to use various solvents for maximum efficiency, e.g., an aqueous mixture of methanol, ethanol and acetone; only methanol; only ethanol; or their aqueous solutions. As noted by Oroian and Escriche, ethanol is the most frequently used solvent for extraction.” (Karoline, 2020)
“Although methanol is cheaper, ethanol is nontoxic and has GRAS status, which means that ethanol can be used safely in food. In the case of methanol or other solvents, their residues must be removed from the extract. For example, the maximum residue limit in extracted foodstuffs or food ingredients for methanol is 10 mg/kg, and for methyl acetate is 20 mg/kg (in coffee or tea); in the preparation of flavorings from natural materials, the maximum residue limits in foodstuffs resulting from the use of extraction solvents are 1 mg/kg for methyl acetate, 2 mg/kg for diethyl ether and 0.02 mg/kg for dichloromethane. Oroian and Escriche also highlighted techniques for extract purification and isolation (e.g., ultrafiltration or macroporous adsorption resins). It should be noted that other extraction processes are available, such as subcritical water extraction, pulsed electric field, ultrasonics, microwave‐assisted extraction, supercritical fluid extraction, solid phase extraction and pressurized liquid extraction. Each of these methods has advantages and disadvantages; however, the purpose of this article is not a detailed description of extraction techniques, and hence they are mentioned only.” (Karoline, 2020)
“It is noteworthy, however, that the efficiency of extracts not only depends on the solvent and extraction techniques, but also on the raw material. For example, Sulas et al. analyzed the antioxidant capacity of Chrysanthemum coronarium L (garland) in two different phenological stages—vegetative and flowering. The extract from the herb at the flowering stage is characterized by a higher antioxidant capacity. Moreover, the authors also found differences between plant organs: stems and heads showed lower antioxidant capacities than leaves. Elwekeel, Elfishway and AbouZid also Antioxidants 2020, 9, 711 9 of 31 noted differences in the content of silymarin depending on the maturity stage of milk thistle (Silybum marianum) fruits; the highest silymarin amount was found in fully mature fruits. Differences in antioxidant ability and polyphenol composition were also found between herbs belonging to the Epilobium genus (Onagraceae). Authors found that E. pariviflorum presented the highest antioxidant capacity from all five examined species, and E. angustfolium was characterized by the most varied and richest composition of the selected herbs. Ghasemi et al. noted that the climatic and geographical conditions of regions of Iran influenced the flavonoid and phenol contents and antioxidant activity of green husk extracts from walnut (Juglans regia L).” (Karoline, 2020)
“In addition to extracts, the product itself should also be considered. Various meat product types exist: hams, cooked sausages, fresh sausages, fermented sausages, air‐dried meat products, liver pâté, patties, nuggets, etc. Each of these has its own specifications and characteristics which translate into safety, shelf life and general quality. For example, cooked sausages, due to their high water activity, are perishable; in contrast, cured and air‐dried products are considered to be more stable due to their production process (drying). The recommended storage temperature for nuggets is around −20 °C, and for spreadable liver sausage is between 0 and 2 °C. In addition, the composition of individual meat products is different; these can be produced from various meat types and cuts, and various spice mixtures can also be applied. All of these factors should be taken into account when experimenting with possible measures to reduce nitrate.” (Karoline, 2020)
Karoline (2020) then moved on to the matter of consumer acceptance. It is a cautionary which we will do very well to take notice of at this stage. “Based on the above outline, reducing nitrate in meat products would appear to be the correct approach. However, this should be associated with the consumer’s desire to buy such a product. An alternative for nitrite could be identified and new meat products could be created, but it remains to be seen whether this would be sufficient for consumers to consider making a purchase. According to Hung, de Kok and Verbeke, consumers are interested in meat products with lower nitrite/nitrate amounts, finding them to be “more natural”.” (Karoline, 2020)
“However, the authors also noted that decisions about the purchase of new meat products are associated with an awareness of the purpose and function of nitrate/nitrite, their reason for application, and their consequences. Thus, a successful reduction of added nitrite in meat products, together with plant fortification, would result in a number of benefits to customers, such as a decrease in cancerogenic compounds in products, in addition to an increase in bioactive compounds.” (Karoline, 2020) This is a point we made before. The approach should not just be to reduce nitrites and nitrates, but to enhance the overall value and quality of the resultant product. In the project questions I proposed to answer, question 8 asks “what conditions must exist to change the status of cured meats (and nitrites in particular) from “possibly cancer-causing” to healthy and desirable!”
They move to the matter of cost, which is one of the reasons for my approach of “root-to-tip” as it generally leads to a reduction in the cost of the plant material and an increase in the income for the farmer, leading to a win-win situation for all parties, above all the consumer who has healthier purchase options. Karoline (2020) remarked that “the cost of plant additives should be considered. Compared to other methods of preparation, freeze drying appears to be more expensive than spray drying or convective drying. However, previous research has noted that the final costs of each method comprise various components costs (e.g., in the case of freeze drying, sublimation of ice, freezing, bound water elimination and total energy consumption). Cost considerations pose another challenge to the reduction of nitrite in meat.” (Karoline, 2020)
CHR Hansen’s System
With regards to plant based fermentation brines, the first serious contender for a complete system must be CHR Hansen who pioneered a re-discovery of the system in 1977 using staphylococci as starter culture.
-> Background
They write about the initial launch of the first commercial form of Staphylococci starter culture in 1977 “the product Bactoferm C-P-77 for cured ham was launched. The idea was to use the positive effect of nitrate reductase activity of the Staphylococci also in cooked ham and emulsified cooked sausage. Unfortunately, this idea was not supported and fizzled out after a while.” (CHR Hansen)
“By coincidence, the idea got a renaissance some years later. Producers of “white sausages” such as “Münchner Weißwurst“ or “Nürnberger Bratwürstchen” had occasional problems with a red centre of the sausages – in particular when there was more time between stuffing and heat treatment than usual. Analyzes showed that also in “white sausages”, especially in recipes with added herbs, significant amounts of nitrate were found. In sausages with a red centre, there was nitrite found as well. Microbial analyzes of such sausages showed an exceptionally high amount of Staphylococci before reaching 45°C in centre. All those observations and the growing scepticism of the use of nitrite as well as the declaration of nitrite as preservative led to the idea to utilize the reddening effect of “white sausages”.” (CHR Hansen)
-> Inclusion rate and method
Their plant-based ingredient used in literature is spray dried Swiss juice chard due to its neutral taste and the absence of allergens. Swiss chard juice contains approximately 3 – 4 % nitrates. They found that when approximately 0.2 % of spray-dried celery or Swiss chard juice powder is used, it results in around 70 ppm of nitrate in the final product.
The French flavour house, Diana Naturals (Tel. +33 299 98 40 77) is one supplier of spray-dried Swiss chard. The starter culture is sold by CHR Hansen as Bactoferm CS-300.
Subsequent to the launch of CS-300, CHR Hansen launched two new products, Rosa and Scarlet. These new products are similar to CS-300. Rosa and Scarlet are a little more active at temperatures of 8-15°C. Above 15°C all three are very similar. I wonder if Rosa refers to dog rose which would mean that they added plant extracts to bolster the colour. If so, it is ingenious!
CH Hansen says the following regarding their targeted approach to different product classes, “Regarding offerings from Chr Hansen we do have cultures targeting bacon where functionality include improvement of colour and flavour, but the desired colour is still depending on some nitrate (or nitrite) being present in the brine, i.e. from natural plant sources.” For bacon they recommend Roxanne LC or Scarlet + B-LC-20. The LC is for bioprotection to make the product safer with less variation from batch to batch, because of better control of the contamination flora.
->> Cooked ham
In the ingredients and process description, I retain their first commercial starterculture just as an example.
->>> Brine Composition
- Water
- Sodium Chloride – standard inclusion rate
- Swiss chard juice powder – 0.2% of total weight
- Bactoferm CS-300.
->>> Processing Steps
Add Bactoferm CS-300 first to the water.
Add Swiss chard juice or whatever plant-based material is used.
Disperse completely.
Add rest of brine ingredients.
Do NOT leave brine overnight!
No long resting period is required. Heat treat during smoking for 2 hours at 45°C followed by the standard heat treatment.
The cooking cycle does not begin at 45 deg C. Delta-T-cooking has a positive effect on color formation. “A pretreatment of two hours at 45°C does not result in a two-hour longer heating process. The time frame between 10°C and 45°C is important for the effect of Bactoferm CS-300. This interval is longer in the center than at the rim of the product. If the starter culture is inactivated/killed early in the process, it will be visible as a pale or even grey rim of the product. The risk of the too early killing of the starter culture is smaller in hams cooked in molds, because of the prolonged temperature transfer.” (CHR Hansen)
->> Bacon and Raw Cured Ham
The process is the same as for Cooked Hams as given above. The challenge is to ensure the plant-based “nitrate carrier” is dispersed to the centre of the meat, especially in larger cuts. This requires increased tumbling time or a resting step which I have always experienced as beneficial for proper diffusion. It is important to remember that most of the diffusion early on in bacon production happens during the heating step in par-cooked bacon products.
Benefits of Beet as Functional Meat Ingredient
-> Colour Development
The question is if there is any real benefit to colour development apart from the obvious source of nitrate and therefore nitrite to effect curing. I know that lower pH products like salami favour colour development from plant-based extracts other than the nitrate/ nitrite pathway. Interesting studies have been done here at the University of Stellenbosch. In my own work, I have never had great results in colour development in hams from plant-based extracts that I did.
Karoline (2020) discussed this matter in great detail. I highlite the studies of interest with a bullet point. These will form a great basis for future work. “An increase in the value of a yellowness (b*) parameter was observed with an increasing amount of PEP added, which was not reported for spinach extract. The authors concluded that the increase in yellowness (b*) was due to the presence of plant pigments.
- Horsch et al. noted that the addition of celery (either in the form of plain concentrate or concentrate with citric acid supplemented) did not affect the redness of the product; the (a*) value was similar to that of the samples with the addition of sodium nitrite. However, they recorded changes in the lightness and yellowness parameters with an increasing amount of celery concentrate—pork ham was found to be more yellow and darker than the control sample.” (Karoline, 2020)
- In another study, the authors compared the effect of young radish and commercial vegetable powder (250 g/kg) on various parameters of cooked pork sausage. According to their results, the powders created darker and less red products than sausage with the addition of sodium nitrite. However, the redness value (a*) for samples with young radish added was greater than that of the product with a vegetable powder addition .
Khaleghi et al. [70] observed that barberry extract decreased the lightness value (L) of beef sausages compared to a control sample to which 120 mg/kg of nitrite was added. The authors also pointed out that the combination of 90 mg/kg plant extract with nitrite not only negatively affected the quality of the meat
product by increasing the formation of thiobarbituric acid reactive substances (TBARS), but also decreased the redness of the product over time.
- The addition of red wine (7.5%) to chouriço (dry-cured sausage) did not change the value of the redness parameter (a*), which recorded the same high value as achieved with the addition of sodium nitrite (150 ppm). The authors also observed that red wine (7.5%) in combination with garlic (1%) increased the yellowness (b) of the product, which was thought to occur because of the interaction between the two ingredients [71].
Similar results were obtained for beef sausages treated with grape pomace (1% and 2%) combined with nitrite (30 mg/kg). In this case, grape pomace lowered the values of the redness (a) and lightness (L) parameters of beef sausages (in comparison to the control sample with the addition of 120 mg/kg nitrite) [72].
- Moawad et al. [73] reported that nitrite (100 or 125 mg/kg) in combination with green tea catechins (300 mg/kg) protects raw cured sausage from pigment deterioration. They recorded the highest values for the b* parameter after the addition of catechins. Aliyari et al. [74] reported that samples (beef sausage) with no added nitrite but with the addition of the highest quantities of extracts of pomegranate peel (Malase torshe saveh variety) and pistachio green hulls (Ahmad aghaei variety) (1250 ppm) exhibited more yellowness (b) than the control sample with nitrite addition (120 ppm). The lightness (L) value of beef sausages increased with an increasing amount of pistachio green hull extracts (from 250 to 1250 ppm with the simultaneous reduction of nitrite from 100 to 0 ppm).
However, the value of the a* parameter decreased in almost all of the treated samples. This approach could also be applied to plant wastes (e.g., fruit peel). Manihuruka et al. [75] suggested the use of red dragon fruit (Hylocereus polyrhizus) peel as a natural colorant and antioxidant. Although the peel contains betacyanins, the redness of beef sausage was found to be less intense, which could be due to the degradation of betacyanins after the boiling process (60–65 °C, 60 min.). They also found an increase in the value of yellowness (b) with increasing quantities of dragon fruit peel extract added in beef sausages, which they attributed to the presence of betaxanthin.
- The addition of 0.2% of adzuki bean extract in the preparation of uncured pork sausage was found to increase the redness of the product, with a simultaneous reduction in the lightness value [76]. Similar results were obtained for pork sausage prepared with various proportions of annatto seed powder (0.025%, 0.05%, 0.1% and 0.2%).
- Another study showed that, with increasing amounts of Bixa orellana (L) seed powder, the values of a* and b* parameters increased and the L* value decreased. It is notable that nitrite levels were reduced in the sample to 37.5 ppm in comparison to the control sample (150 ppm) [77].
Çemtekin et al. [78] reported that there were no significant differences for the L* parameter when they tested samples (turkey meat model system) following the addition of Viburnum opulus (L) and Crataegus monogyna, and after 30 days of storage under aerobic and anaerobic conditions. They obtained differences in values of the a* parameter, which increased with an increasing quantity of V. opulus. Lin et al. [79] tested the effects of different concentrations of green tea extract (GTE; 0.02%, 0.05%, 0.1%, 0.2% and 0.5%) in pepperoni, and obtained the highest values of color (L, a, b) parameters for salami treated with 0.05% GTE in combination with 0.009% nitrite.
- Vossen et al. [80] tested the effect of dog rose (Rosa Canina L.) fruit extract on the color parameters of frankfurters. At the end of the tested storage period, there were slight differences observed in the values of the color parameters; the value of yellowness had increased and the addition of a greater quantity of the extract increased the value of the a* parameter.
- Hayes, Canonico, and Allen [81] studied the effect of tomato pulp powder in luncheon roll products, and found that the values of redness (a) and yellowness (b) increased and the value of lightness (L) decreased.
- Another group of researchers studied the effects of grape seed extract with olive pomace hydroxytyrosol, and chestnut extract with olive pomace hydroxytyrosol, on the color values of Cinta Senese dry‐fermented sausage. The values of redness were increased by the addition of grape seed extract, which the authors concluded was probably an effect of the formation of Zn‐protoporphyrin [82].
- Shin et al. [83] reported that 2% preconverted nitrite from Swiss chard powder positively affected the formation of nitrosoheme pigments in cooked pork patties. Swiss chard powder was also tested in combination with 60 ppm of nitrite, which resulted in satisfactory results for the a* parameter obtained.
- Sucu and Turp [84] observed an increase in the values of the redness parameter in sucuk with the addition of beet (Beta vulgaris) extract. They obtained the highest values from the sample containing 0.35% of the beet extract. They also noted that beet extract decreased the value of yellowness (b) and increased the value of lightness (L) during the first few days of storage.
Jin et al. [85] studied the effects of various plant extracts and powders, and recorded a positive effect on the CIE Lab* parameters in pork sausage; the predominant effect was an increase in the values of the redness (a) parameter. Fruits are not rich in nitrates [11], and due to a lack of information about nitrite substitution using plant extracts, scientists have investigated extracts from karanda (Carissa carandas Linn), sour cherry (Prunus cerasus L.) and blackcurrant (Ribes nigrum L.) leaves [86,87]. Karanda is a herb from Thailand that is used in folk medicine to treat intermittent fever, earache and oral inflammation. The dark violet fruits of this plant are rich in anthocyanins and contain high amounts of vitamin C. However, anthocyanins do not improve the color of meat products. Researchers have studied the effect of anthocyanins after 36 h of fermentation of sausages, and found that the color of the fermented sausages had higher values of yellowness (b*) and lightness (L) parameters compared to the control sample with nitrite ddition [86]. Nowak et al. obtained similar results [87]. They observed that P. cerasus and R. nigrum did not increase the value of the redness (a*) parameter of pork sausage, and that the extracts were responsible for the high value of the yellowness (b*) parameter. Researchers are also evaluating the possibility of using vacuum‐packed extracts to preserve the qualities of meat products. Marazzeq et al. [88] reported that it is not possible to completely replace nitrite with olive leaf extract (OLE). They conducted various trials with different combinations of OLE and nitrite, and found that with decreasing levels of synthetic additives, there was a decrease in the redness parameter. The sample with total nitrite replacement demonstrated the lowest value of the redness parameter after 12 weeks of storage.
- According to Nowak et al. [87], with the application of plant extract, it is necessary to include a natural color component during the preparation of the meat product.
-> Betalains
Betalains are the colour component in Beetroot. The value has been touched upon in the previous section and it is sensitive to cooking and pH. I nevertheless include this short section by Goldman & Janick (2021) as knowing what it is all about is beneficial. I also list three studies to be looked at further at the end of this section as we consider their inclusion in meat formulations. I also list it in the Further Research section.
“Beets possess vibrant red and yellow pigments that include the red-violet betacyanins and the yellow betaxanthins (Clement et al., 1992). Betalains are produced from the amino acid tyrosine, and Lopez-Nieves et al. (2017) suggested that relaxed sensitivity to tyrosine inhibition may explain the evolution of betalain biosynthesis. Betalains exhibit health properties such as antioxidant activity (Tesoriere et al., 2009) and the induction of Phase II enzymes that may protect against certain cancers (Lee et al., 2005). Betalains are controlled by two loci in beet, R and Y (Goldman and Austin, 2000). Beet roots are red when dominant alleles are present at these two loci, and roots are white when alleles at the Y locus are recessive. When the R locus is homozygous recessive but alleles at Y are dominant, the root is yellow (Hatlestad et al., 2012). Red beets typically have five to eight times more betalains than yellow beets because the conversion of tyrosine to betacyanin is far more efficient than that for betaxanthin (Wang et al., 2017). Betalain concentration for both betacyanin and betaxanthin has been increased in table beet through recurrent selection (Gaertner and Goldman, 2005). The dried, powdered product for table beet roots is used as a commercial food dye.” (Goldman & Janick, 2021)
When considering betalains in meat processing Ceclu (2020) offers caution when he states:
- Betalains are susceptible to pH, oxygen, metal ions, temperature, water activity, exposure to light and enzymatic activities (1 and 2 below);
- High temperature, pH changes or enzyme presence could convert betanin to betanidin.
In this regard, three works must be consulted (3 below):
- Herbach KM, Stintzing FC, Carle R (2006) Betalain stability and degradation Structural and chromatic aspects. Journal of Food Science 71: 41-50.
- Sekiguchi H, Ozeki Y, Sasaki N (2013) Biosynthesis and regulation of betalains in red beet. In: B Neelwarne, Red beet biotechnology – food and pharmaceutical applications. Springer Science+Business Media, New York, 45-54.
- Wiczkowski W, Romaszko E, Szawara-Nowak D, Piskula MK (2018) The impact of the matrix of red beet products and interindividual variability on betacyanins bioavailability in humans. Food Res Int 108: 530-538.

Conical beet roots in the 17th-century painting “A fruit stall” by Snijders (see inset). Source: The Bowes Museum, Barnard Castle, County Durham, United Kingdom.
-> Antioxidants
Karoline (2020) did an excellent review of the antioxidative properties of various plants which is a very good reason for including plant matter in meat formulations. I again highlite the work of interest to us. They begin with a great overview of the process and importance of antioxidants. They write, “The method of protective effect imparted by nitrite against the oxidation process in cured meat products is similar to that of protecting the discoloration of the meat product. In meat products, free ferric ions can act as a catalyst of oxidation. After curing, NO binds to iron from heme and inhibits the process of lipid peroxidation, thereby enhancing product stability during storage [64,65]. In addition, S‐nitrosocysteine is formed due to the utilization of oxygen during the oxidation of NO to NO2 [40,64].”
“Plant extracts demonstrate strong antioxidant activities [89]. Polyphenols can inhibit the propagation of free radicals and their formation through iron chelation [66]. Sueprasarn et al. [86] conducted studies on the effect of karanda extract (0.05% w/w concentration) on the storage of Nham sausage. According to their results, karanda extract did not inhibit the process of lipid oxidation during 30 days of storage, and its activity was similar to nitrite. There was a significant increase in the value of TBARS in fermented sausage, which could be a result of the degeneration of antioxidant chemicals through light and heat exposure. Nowak et al. [87] used extracts from sour cherry and blackcurrant leaves, and found that the reduction of malondialdehyde (MDA), after 14 and 28 days of storage, was similar to that of nitrite. The leaves of R. nigrum show a stronger antioxidant activity than those of P. cerasus, which might be attributed to the higher amounts of polyphenolic compounds present in the leaves. Kim et al. [66] demonstrated that the amount of plant extract applied is very important in inhibiting the formation of TBARS in meat products. In their experiments, they recorded lower values of MDA in samples with the addition of the highest quantity of fermented spinach extract, compared to the values of the control sample. Kurćubić et al. [89] also showed that the concentration of the plant extract is an important factor in maintaining the quality of the meat product. They conducted experiments with two samples using extracts of Kitaibelia vitifolia of 30.0 (concentration 3% w/v) and 12.5 (concentration 10% w/v) g per kg of meat dough. According to their results, the application of 10% concentrate at a lower dose provided greater protection than samples with 3% concentrate applied at a higher dose. In general, K. vitifolia extract showed greater antioxidant activity than nitrite. Another study showed that higher quantities of barberry extract resulted in better antioxidant activity during the first few days of storage, but this effect decreased over time. The authors suggested that there was an antagonistic effect between phenolic compounds of barberry extract and nitrite [70]. A similar observation has been reported with regard to TBARS—two compounds at higher quantities work in an antagonistic way [70]. Sucu and Turp [84] observed similar results in sucuk after testing the extract of beet. They observed that although B. vulgaris contains polyphenolic compounds, they did not decrease the TBARS values during storage. Riazi et al. [72] reported that higher quantities of natural antioxidants do not lead to a reduction in the formation of MDA. However, Moawad, Abozeid and Nadir [73] reported that 300 mg/kg of green tea catechins in combination with 100 or 125 mg of nitrite added to dry‐fermented sausage shows stronger antioxidant properties than those of control samples. Lin et al. [79] reported that although the addition of GTE (0.05%) to salami contributes to the lowering of TBARS values, its combination with nitrite (0.009%) is not effective. As previously suggested, the combination of both plant extract and nitrite might result in a pro‐oxidant status. For example, Hayes et al. [81] reported a pro‐oxidant effect of nitrite and tomato pulp powder in luncheon roll. Jin et al. [85] also recorded an increase in the value of TBARS in samples prepared with various plant extracts. However, Shin et al. [83] reported the synergistic effects of nitrite and plant extract against lipid oxidation in pork patties. Ko, Park and Yoon [69] demonstrated that vegetable powders (young radish and a combination of cabbage and Chinese cabbage) can prevent lipid oxidation in cooked pork sausages during storage of up to 20 days. Moarefian et al. [90] reported that after 30 days of storage, all sausages treated with various quantities of the essential oil (EO) of Mentha piperita combined with nitrite provided satisfactory results. According to their results, 20 ppm EO and 100 ppm of nitrite added to the product demonstrated the strongest lipid oxidation inhibition activities, compared to 120 ppm of NaNO2 alone. Adzuki bean (Vigna angularis) is a rich source of catechins, anthocyanins and chlorogenic acid. Jayawardana et al. [76] studied samples prepared with Adzuki bean extract (0.2%, 0.3%, 0.4% and 0.5%) and found that the extract showed a good inhibitory effect of lipid oxidation, which was similar to that of 0.1% BTH. The authors concluded that 0.2% of the extract was
optimal to inhibit the lipid oxidation process in comparison to the product prepared with 0.008% sodium nitrite alone. Çemtekin et al. [78] noted that guelder rose (V. opulus L) and hawthorn (C. monogyna) decreased the amount of MDA formed in cooked turkey during the first 30 days of storage under aerobic and anaerobic conditions. The authors noted that the highest quantities of the plant extracts showed the greatest inhibitory effect toward the formation of TBARS. Similar observations were recorded by van Cuong and Thoa [77]. They added the seed powder of B. orellana (L) to the product, which contributed to the reduction of primary and secondary lipid oxidation products to an extent similar to that achieved in the control sample (150 ppm nitrite) [77]. Another study demonstrated that the antioxidant capacity of the product increases with an increasing quantity of plant extract. For example, H. polyrhizus peel extract added to the product increased antioxidant activity from 49.71% (control sample) to 54.76%, 60.89% and 72.94%, respectively, with 20%, 30% and 40% of the extract. Similar results were obtained for the TBARS parameter [75]. Another study reported the use of extracts of pomegranate peels and pistachio green hulls to decrease the formation of MDA [74]. Sharma et al. [91] found the addition of 5000 ppm of turmeric powder to be the most effective for slowing down the process of lipid oxidation.
This review provides fertile information for the design of a brine. The antioxidative properties of plant material are without a doubt a reason for its inclusion in meat formulations. The work of Prof Tappel and his research teams must be factored in and a multi-plant strategy must be developed.
-> Phytochemicals
Back to beetroot, in particular, let’s say something about the presence of phytochemicals which has an effect on the colour and antioxidative properties. Phytochemicals is a class of chemicals found in beetroot which must be investigated in great detail. There is an amazing volume of recent work done on it, but there are enough benefits demonstrated for the consideration of beetroot than to go into too much detail about phytochemicals at this time. “The phytochemicals that give veg their colour are understood to be beneficial. Several of these phytochemicals are antioxidants that may protect our bodies from the presence or actions of harmful free radicals.” (lovebeetroot.co.uk)
-> Anti-Microbial Efficacy of Plant Extracts
We lastly look at the antimicrobial efficacy of plant extracts, again by using the short overview done by Karoline (2020).
“Nitrite also shows strong microbicidal activity against some foodborne pathogens, such as Clostridium botulinum, Listeria monocytogenes and Escherichia coli. The bacteriostatic property of nitrite in cured meat products is associated with the formation of NO and/or HNO2. Researchers hypothesize that the bactericidal activity results from the inactivation of iron–sulfur proteins of the bacteria by the presence of NO in the meat product. The bactericidal activity of nitrite is affected by various factors, such as salt concentration, heat treatment, various curing ingredients and pH value [64,65]. Plant extracts contain various compounds (e.g., phenolics, flavonoids, tannins and saponins) that show strong antimicrobial activity, which is related to the presence of the hydroxyl group (‐OH). The mechanism of action of phenolic compounds is based on disrupting the structure of the proteins present on the bacterial cell wall. This results in the leakage of the cellular components which kills the bacteria [92]. Riel et al. [67] added parsley extracts to mortadella‐type sausage. According to their results, with an increase in the amount of the extract, the bacterial cell count of L. monocytogenes decreased. The researchers concluded that a higher amount of vegetable extract causes higher nitrate and, therefore, nitrite formulation; examination proved that the greatest inhibition properties were in samples with the highest extract addition (4.29 g/sausage meat). Ko, Park and Yoon [69] suggest the possibility of applying vegetable powders (especially from young radish) as a natural source of nitrite for microorganism inhibition. Furthermore, celery concentrate (75.6 g) inhibited the growth of L. monocytogenes in a manner similar to that achieved by 100 mg/kg of sodium nitrite. Similarly, pork ham treated with 151.2 g of celery concentrate inhibited the growth of L. monocytogenes in a manner similar to achieved by 200 mg/kg sodium nitrite. However, the effect of celery concentrate was most effective in combination with 10% citric acid [68]. Xi et al. [93] observed that the addition of natural powders or extracts to pork sausage (meat model system) resulted in the inhibition of growth of L. monocytogenes. Cranberry powder, GTE, grape seed extract and cherry, lemon and lime powders (in higher quantities) reduced the bacterial count in the product. Bakhtiary et al. [94] reported that EOs in combination with sodium nitrite (100 or 200 mg/kg) synergistically inhibited the growth of Clostridium sporogenes and C. perfringens. The authors reported that EOs from Satureja bachtiarica Bunge showed the strongest inhibitory activity in beef fillets inoculated with Clostridium species during storage of up to 30 days. The antimicrobial effect of EOs is mainly associated with their chemical composition [94]. Another study analyzed the counts of E. coli, Salmonella sp. and S. aureus and found that beef sausages had bacterial counts below 3 log 10 cfu/g [75]. The authors attributed this efficiency to good hygienic practice during the production process. Shin et al. [83] reported that there were no coliform bacteria detected in pork patties. Patarata et al. [71] noted that red wine in combination with garlic showed antibacterial activity against C. sporogenes and Salmonella in dry‐cured sausages—chouriços. Similar results were obtained by Aquilani et al. [82]. They used grape seed and chestnut extract in the preparation of Cinta Senese dry‐fermented sausage and found that the counts of E. coli, L. monocytogenes, Clostridium spp., Salmonella spp. and Staphylococcus spp. were low for up to 24 days of ripening. The authors attributed the efficiency of these extracts to the phenolic content. The addition of 1.56% winter savoy (S. montana) EO inhibited the spore formation of C. perfingens in mortadellatype sausage. [95]. Similar observations were made by Khaleghi et al. [70]. The authors noted that the TVC was particularly low for samples with the addition of 90 mg/kg barberry (B. crataegina) extract and nitrite (30 or 60 mg/kg). In addition, C. botulinum was not detected in samples during 30 days of storage, including in samples with no nitrite or plant extract added. Gyawali and Ibrahim [92] indicated that variations in the chemical and structural composition of phenolic compounds may result in differences in the antimicrobial properties of plant extracts. As Nowak et al. [87] reported, despite the high polyphenolic content of leaf extracts of P. cerasus and R. nigrum, their addition to the pork sausages did not stop the growth of psychotrophic and mesophilic bacteria after 28 days of storage. However, according to the literature, only Pseudomonas spp. were inhibited by leaf extracts during the entire storage period. Van Cuong and Thoa [77] found that the total count of bacteria and Enterobacteriaceae was lower in all samples with the addition of annatto seed powder, regardless of its quantity. However, the effect was similar to nitrite. The addition of extracts from K. vitifolia to dryfermented sausage helped in decreasing the count of harmful microorganisms (e.g., P. vulgaris, Bacillus subtilis and Aspergillus niger). The strongest inhibitory effect was seen against E. coli [89].” (Karoline, 2020)
So much work has been done in this field over many years that I leave it here by way of introduction. One could spend years only on this subject.
-> Toxicity
Investigate the following Toxicity report by James A. Duke. 1983. Handbook of Energy Crops. unpublished.
“Feeding sugar beet to sheep has caused renal calculi, composed of uric and phosphoric acids with lime. Fresh leaf may also cause poisoning due to the 1% oxalic acid therein. Leaf may also contain dangerous levels of HCN and/or nitrates and nitrites. Betaine acts as a mild diuretic. Beet pollen can cause hay fever. Sugar appears to have caused dermatitis in two-thirds of the workers in one crystallizing department.”
-> Folk Medicine
List folklore on Beetroot, also listed by James A. Duke. 1983. Handbook of Energy Crops. unpublished.
-> Studies of benefits to human health
The work of Godofredo U. Stuart, Jr.’s listed under benefits to human health and the references under “References” must be studied and incorporated into the overall work on Beetroot in the future.
-> C Botulinum in meat products
Related to the general safety of plant-based curing, CHR Hansen alerted me to two important studies to consider namely:
- Project no. 18550, doc. no. 37215.3, from 26. October 2006 the Danish Meat Institute indicated that “the worries about the germination and growth of Clostridia spores could not be validated as cooked hams or emulsified sausages are usually stored below 7°C for approximately 1-2 months only.” (CHR Hansen)
- F.-K. Lücke, who came to the came to the same conclusion in 2003 as the Danish Meat Institute. (document 160 , “Mitteilungsblatts der Bundesanstalt für Fleischforschung”, Kulmbach, p. 95-104).
-> Future studies with sea beet to discover its likely uses in pre-history
I would, at a future time, get sea beet to examine its uses in pre-history. It can be found along the German coast as per the following work. Sarah Driessen, Matthias Pohl, Detlef Bartsch. (2021) RAPD-PCR analysis of the genetic origin of sea beet (Beta vulgaris ssp. maritima) at Germany’s Baltic Sea coast, Basic and Applied Ecology, Volume 2, Issue 4, 2001, Pages 341-349, ISSN 1439-1791, https://doi.org/10.1078/1439-1791-00061. (https://www.sciencedirect.com/science/article/pii/S1439179104700560)
-> Betalains in Meat formulations
When considering betalains in meat processing Ceclu (2020) offers caution when he states:
- Betalains are susceptible to pH, oxygen, metal ions, temperature, water activity, exposure to light and enzymatic activities (1 and 2 below);
- High temperature, pH changes or enzyme presence could convert betanin to betanidin.
In this regard, three works must be consulted (3 below):
- Herbach KM, Stintzing FC, Carle R (2006) Betalain stability and degradation Structural and chromatic aspects. Journal of Food Science 71: 41-50.
- Sekiguchi H, Ozeki Y, Sasaki N (2013) Biosynthesis and regulation of betalains in red beet. In: B Neelwarne, Red beet biotechnology – food and pharmaceutical applications. Springer Science+Business Media, New York, 45-54.
- Wiczkowski W, Romaszko E, Szawara-Nowak D, Piskula MK (2018) The impact of the matrix of red beet products and interindividual variability on betacyanins bioavailability in humans. Food Res Int 108: 530-538.
-> Anti-inflammatory activity of betalains: A comprehensive review
-> Current Knowledge on Beetroot Bioactive Compounds: Role of Nitrate and Betalains in Health and Disease
-> Nutritional, Bioactive and Physicochemical Characteristics of Different Beetroot Formulations
Further Reading
Beta maritima. The origin of beets: buy book and study
References
Edward Abbott, (1864) The English and Australian Cookery Book. Cookery for the Many, as well as for the “Upper Ten Thousands.” By an Australian Aristologist. London: Sampson, Low, Son and Marston, 14, Ludgate Hill.
Added Sugars, American Heart Association
Muhammet İrfan Aksu, Ebru Erdemir, Emre Turan, İhsan Güngör Sat. (2020) Effects of red beet extracts on protein and lipid oxidation, colour, microbial, sensory properties and storage stability of Turkish pastırma. Journal of Stored Products Research, Volume 89, 2020, 101721, ISSN 0022-474X, https://doi.org/10.1016/j.jspr.2020.101721. (https://www.sciencedirect.com/science/article/pii/S0022474X20304665)
Boroujerdnia M, Ansari NA, Dehcordie FS. Effect of cultivars, harvesting time and level of nitrogen fertilizer on nitrate and nitrite content, yield in Romaine lettuce. Asian J Plant Sci. 2007;6(3):550–3. [Google Scholar]
Bednar C, Kies C. Nitrate and vitamin C from fruits and vegetables: Impact of intake variations on nitrate and nitrite excretions of humans. Plant foods for human nutrition. 1994;45(1):71–80. 10.1007/bf01091231 [PubMed] [CrossRef] [Google Scholar]
Bill H. To: HEYDENS, WILLIAM F [AG/1000] Subject: FW: summary article.
Ceclu L, Oana-Viorela N (2020) Red Beetroot: Composition and Health Effects – A Review. J Nutri Med Diet Care 6:043. doi.org/10.23937/2572-3278.1510043
CHR Hansen: Pamphlet – Natural curing without added nitrite and personal correspondence, 2021 and 2022.
EFS. Nitrate in vegetables-Scientific Opinion of the Panel on Contaminants in the Food chain. EFSA Journal. 2008;6(6):689. [Google Scholar]
Ellet, E. F. (Elizabeth Fries), 1818-1877: The new cyclopaedia of domestic economy and practical housekeeper: adapted to all classes of society and comprising subjects connected with the interests of every family, such as domestic education, houses, furniture, duties of mistress, duties of domestics, the storeroom, marketing, table and attendance, care and training of children, care of the sick, preparation of food for children and invalids, preservation of health, domestic medicine, the art of cookery, perfumery, the toilet, cosmetics, and five thousand practical receipts and maxims: from the best English, French, German, and American sources: illustrated with over two hundred engravings / (Norwich, Conn.: Henry Bill Pub. Co., 1872) (page images at HathiTrust)
FSA. (2008) Nitrate in vegetables Scientific Opinion of the Panel on Contaminants in the Food chain. The EFSA Journal (2008) 689, 1-79.
Goldman, Irwin L. and Janick, Jules (2021). Review – Evolution of Root Morphology in Table Beet: Historical and Iconographic; Frontiers in Plant Science; https://doi.org/10.3389/fpls.2021.689926
Hill MJ. Nitrates and nitrites in food and water: CRC Press; 1996. [Google Scholar]
Hmelak Gorenjak A, Cencič A. Nitrate in vegetables and their impact on human health. A review. Acta alimentaria. 2013;42(2):158–72. [Google Scholar]
Huarte-Mendicoa J, Astiasaran I, Bello J. Nitrate and nitrite levels in fresh and frozen broccoli. Effect of freezing and cooking. Food Chemistry. 1997;58(1–2):39–42. [Google Scholar]
Karolina, F., Wójciak, K. M.. (2020) Reduction of Nitrite in Meat Products through the Application of Various Plant‐Based Ingredients. MDPI, Antioxidants.
Kale RG, Sawate AR, Kshirsagar RB, Patil BM and Mane RP. (2018) Studies on evaluation of physical and chemical composition of beetroot (Beta vulgaris L.); International Journal of Chemical Studies 2018; 6(2): 2977-2979; P-ISSN: 2349–8528 E-ISSN: 2321–4902 IJCS 2018; 6(2): 2977-2979 © 2018 IJCS
McQueen, A. A.. (1926) Peruvian Public Finance. Trade Promotion Series, Issues 30-37; Department of Commerce, Bureau of Foreign and Domestic Commerce, Julius Klein, Director, compiled by Charles A. McQueen, special agent.
Monakhova YB, Diehl BW. Authentication of the origin of sucrose-based sugar products using quantitative natural abundance (13) C NMR. J Sci Food Agric. 2016 Jun; 96 (8) : 2861-6. doi: 10.1002/jsfa.7456. Epub 2015 Oct 22. PMID: 26362834.
Pavlou GC, Ehaliotis CD, Kavvadias VA. Effect of organic and inorganic fertilizers applied during successive crop seasons on growth and nitrate accumulation in lettuce. Scientia Horticulturae. 2007;111(4):319–25. [Google Scholar]
Pourmoghim M, Khoshtinat K, Makkei A, Fonod R, Golestan B, Pirali M. Determination of nitrate contents of lettuce, tomatoes and potatoes on sale in Tehran central fruit and vegetable market by HPLC. Iranian Journal of Nutrition Sciences & Food Technology. 2010;5(1):63–70. [Google Scholar]
Prasad S, Chetty AA. Nitrate-N determination in leafy vegetables: Study of the effects of cooking and freezing. Food Chemistry. 2008;106(2):772–80. [Google Scholar]
Pretty JN, Brett C, Gee D, Hine R, Mason C, Morison J, et al. An assessment of the total external costs of UK agriculture. Agricultural systems. 2000;65(2):113–36. [Google Scholar]
RAHMANI HR. INVESTIGATION OF NITRATE POLLUTION IN THE SOIL, WATER AND PLANTS IN SAME AGRICL1LTUTAL FIELDS IN BARAAN (ESFAHAN). 2006.
Ramachandran A, Hrycan W, Bantle J, Waterer D. Seasonal changes in tissue nitrate levels in fall-planted spinach (Spinacia oleracea). Website: http://www.usask.ca/agriculture/plantsci/vegetable. 2005.
Ravichandran, P et al. “Optimizing the route for production of activated carbon from Casuarina equisetifolia fruit waste.” Royal Society open science vol. 5,7 171578. 11 Jul. 2018, doi:10.1098/rsos.171578
Rippe, J. M., & Angelopoulos, T. J. (2016). Relationship between Added Sugars Consumption and Chronic Disease Risk Factors: Current Understanding. Nutrients, 8(11), 697. https://doi.org/10.3390/nu8110697
Sadeghi E, Hashemian A, Mohammadi M, Bohlouli Oskoii S, Meskini H, Mohammadi R, et al. Study on the effect of boiling and freezing process on nitrate and nitrite levels in abundant consumed vegetables. Iranian Journal of Nutrition Sciences & Food Technology. 2013;8(3):201–8. [Google Scholar]
Salehzadeh, H., Maleki, A., Rezaee, R., Shahmoradi, B., & Ponnet, K. (2020). The nitrate content of fresh and cooked vegetables and their health-related risks. PloS one, 15(1), e0227551. https://doi.org/10.1371/journal.pone.0227551
Shahlaei A, Ansari NA, Dehkordie FS. Evaluation of nitrate and nitrite content of Iran Southern (Ahwaz) vegetables during winter and spring of 2006. Asian J Plant Sci. 2007;6(1):97–12. [Google Scholar]
The Sugar Beet, Volume 23, Issue 5 (1902)
Tallis, John. (1854) Tallis’s History and Description of the Crystal Palace: And the Exhibition of the World’s Industry in 1851. It was originally published in 1852, and I quote from the reproduction in 1854. It was originally published in three volumes and later combined into one.
Tamme T, Reinik M, Roasto M, Juhkam K, Tenno T, Kiis A. Nitrates and nitrites in vegetables and vegetable-based products and their intakes by the Estonian population. Food additives and contaminants. 2006;23(4):355–61. 10.1080/02652030500482363 [PubMed] [CrossRef] [Google Scholar]
Urbanus BL, Cox GO, Eklund EJ, Ickes CM, Schmidt SJ, Lee SY. Sensory differences between beet and cane sugar sources. J Food Sci. 2014 Sep;79(9):S1763-8. doi: 10.1111/1750-3841.12558. Epub 2014 Aug 14. PMID: 25124655.
Székely, D., Illés, B., Stéger-Máté, M., Monspart-Sényi, J.. (2016) Effect of drying methods for inner parameters of red beetroot (Beta vulgaris L.). Acta Univ. Sapientiae, Alimentaria, 9 (2016) 60-68; DOI: 10.1515/ausal-2016-0006
Tamme T, Reinik M, Roasto M, Juhkam K, Tenno T, Kiis A. Nitrates and nitrites in vegetables and vegetable-based products and their intakes by the Estonian population. Food additives and contaminants. 2006;23(4):355–61. 10.1080/02652030500482363 [PubMed] [CrossRef] [Google Scholar]
Ware, M. (2019); Medical News Today: What are the benefits of beetroot? Medically reviewed by Kathy W. Warwick, R.D., CDE, Nutrition.
Williams GM, Aardema M, Acquavella J, Berry SC, Brusick D, Burns MM, et al. (2016) A review of the carcinogenic potential of glyphosate by four independent expert panels and comparison to the IARC assessment. Critical Reviews in Toxicology. 2016;46(sup1):3–20. 10.1080/10408444.2016.1214677 [PubMed] [CrossRef] [Google Scholar]
Yusof, N. L. (2014). Vacuum Impregnation of Spinach Tissue: Metabolic Consequences and their Potential Industrial Applications. Department of Food Technology, Lund University.
Zhu, Ivan, Getting, Tom. (2012) A review of nitrate reduction using inorganic materials. DOI: 10.1080/09593330.2012.706646; DO – 10.1080/09593330.2012.706646; Environmental Technology Reviews; Taylor & Francis
Godofredo U. Stuart, Jr.’s references.
(1) Hepatoprotective activity of Beta vulgaris against CCl4-induced hepatic injury in rats / M Agarwal et al / Fitoterapia, Feb 2006; Volume 77, Issue 2: pp 91-93 / doi:10.1016/j.fitote.2005.11.004
(2) Effects of chard (Beta vulgaris L. var. cicla) extract on pancreatic B cells in streptozotocin-diabetic rats: a morphological and biochemical study / S Bolkent, R Yanarda et al / Journal of Ethnopharmacology,
Volume 73, Issues 1-2, November 2000, Pages 251-259 / doi:10.1016/S0378-8741(00)00328-7
(3) AN INVESTIGATION OF ANTIMICROBIAL COMPOUNDS FOR IMMUNOMODULATING AND ANTI-ADHESION PROPERTIES / Nafisa Hassan Ali et al / Pakistan Research Repository
(4) Dietary supplementation with leaf extract of Beta vulgaris L. var. benghalensis Hort. in modifying cytotoxicity of lead subacetate in mouse in vivo / Phalguni Nandi et al / Phytotherapy Research, Vol 11 Issue 4, Pages 273 – 276 / DOI 10.1002/(SICI)1099-1573(199706)11:4<273::AID-PTR73>3.0.CO;2-5
(5) Duodenum protecting effects of table beet (Beta vulgaris L. ssp. esculenta var. rubra) during hepatic ischaemia-reperfusion / L Vali et al / Acta Alimentaria, Vol 35, Number 4/December 2006 / DOI 10.1556/AAlim.35.2006.4.8
(6) Anti-Inflammatory Activity of Aqueous Extract of Beta Vulgaris L. / Swati Jain, Vipin Kumar Garg and Pramod Kumar Sharma / Journal of Basic and Clinical Pharmacy
(7) Beta vulgaris L. (accepted name) / Chinese names / Catalogue of Life, China
(8) Beta vulgaris (Beet) / Common names / Zipcodezoo
(9) BETANINE EXTRACTION FROM BETA VULGARIS – EXPERIMENTAL RESEARCH AND STATISTICAL MODELING / Aura STURZOIU, Marta STROESCU, Anicuţa STOICA, Tănase DOBRE / U.P.B. Sci. Bull., Series B, Vol. 73, Iss. 1, 2011
(10) Cytotoxic effect of the red beetroot (Beta vulgaris L.) extract compared to doxorubicin (Adriamycin) in the human prostate (PC-3) and breast (MCF-7) cancer cell lines. / Kapadia GJ, Azuine MA, Rao GS, Arai T, Iida A, Tokuda H. / Anticancer Agents Med Chem. 2011 Mar; 11(3): pp 280-284 / DOI: 10.2174/187152011795347504
(11) PROTECTIVE ROLE OF BETA VULGARIS L. LEAVES EXTRACT AND FRACTIONS ON ETHANOL-MEDIATED HEPATIC TOXICITY / NILESH K. JAIN* and ABHAY K. SINGHAI / Acta Poloniae Pharmaceutica ñ Drug Research, 2012; Vol 69, No 5: pp 945-950 n/ ISSN 0001-6837
(12) Peroxidase production from hairy root cultures of red beet (Beta vulgaris) / Thimmaraju Rudrappa and Bhagyalakshmi Neelwarne* / Electronic Journal of Biotechnology ISSN: 0717-3458 Vol. 8 No. 2, Issue of August 15, 2005
(13) HEMATOPOIETIC STUDY OF THE METHANOLIC ROOT EXTRACT OF BETA VULGARIS ON ALBINO RATS-AN IN VIVO STUDY / INDHUMATHI.T* AND KANNIKAPARAMESWARI.K / Int J Pharm Bio Sci 2012 Oct; 3(4): (B) 1005 – 1015
(14) Phenolics and Betacyanins in Red Beetroot (Beta vulgaris) Root: Distribution and Effect of Cold Storage on the Content of Total Phenolics and Three Individual Compounds / Tytti S. Kujala ,* Jyrki M. Loponen , Karel D. Klika , and Kalevi Pihlaja / J. Agric. Food Chem., 2000, 48 (11), pp 5338–5342 / DOI: 10.1021/jf000523q
(15) Preparation and characterization of Beta vulgaris pulp powder as a pharmaceutical excipient / Pharmacognosy Journal, 2010; 2(11):395–399
(16) Influence of Beta Vulgaris Supplementation on the Level of Oxidative DNA Damage/Oxidative Stress in Head and Neck Cancer Patients Undergoing Radiotherapy / Krzysztof Roszkowski* / J Nucl Med Radiat Ther 2012, 3:4 / http://dx.doi.org/10.4172/2155-9619.100013
(17) ANTIOXIDANT AND ANTI-INFLAMMATORY ACTIVITY OF ETHANOLIC EXTRACT OF BETA VULGARIS LINN. ROOTS / Rita Chakole, Shubhangi Zade, Manoj Charde / International Journal of Biomedical and Advance Research, Vol 2, No 4 (2011)
(18) Effects of chard (Beta vulgaris L. var cicla) on the liver of the diabetic rats: a morphological and biochemical study. / Ozsoy-Sacan O1, Karabulut-Bulan O, Bolkent S, Yanardag R, Ozgey Y. / Biosci Biotechnol Biochem. 2004 Aug;68(8): pp 1640-1648 / DOI: 10.1271/bbb.68.1640 / PMID: 15322346
(19) Evaluation of beetroot (Beta vulgaris L.) leaves during its developmental stages: a chemical composition study / Polyana Batoqui França BiondoI; Joana Schuelter BoeingI; Érica Oliveira BarizãoI; Nilson Evelazio de SouzaII; Makoto MatsushitaI; Claudio Celestino de OliveiraI; Marcela BoroskiI,III; Jesuí Vergílio VisentainerI* / Food Sci. Technol (Campinas) vol.34 no.1 Campinas Jan./Mar. 2014 Epub Mar 11, 2014 / http://dx.doi.org/10.1590/S0101-20612014005000007
(20) EVALUATION OF IN-VITRO ANTI-PROLIFERATIVE ACTIVITY AND IN-VIVO IMMUNOMODULATORY ACTIVITY OF BETA VULGARIS / GITANJALI TRIPATHY*, DEBASISH PRADHAN / Asian Journal of Pharmaceutical and Clinical Research Vol 6, Suppl 1, 2013
(21) Effect of Beta vulgaris extract on triglyceride and cholesterol in diabetic male rats / M Khalili; MR Vaez Mahdavi / Iranian Journal of Pharmaceutical Research (2004): Supplement 2: 55-55
(22) Beta vulgaris / Synonyms / The Plant List
(23) The Potential Benefits of Red Beetroot Supplementation in Health and Disease / Tom Clifford, Glyn Howatson, Daniel J. West, and Emma J. Stevenson* / Nutrients. 2015 Apr; 7(4): 2801–2822. / doi: 10.3390/nu7042801
(24) Beetroot (Beta vulgaris L.) Extract Ameliorates Gentamicin-Induced Nephrotoxicity Associated Oxidative Stress, Inflammation, and Apoptosis in Rodent Model / Ali A. El Gamal, Mansour S. AlSaid, Mohammad Raish, Mohammed Al-Sohaibani, Shaza M. Al-Massarani, Ajaz Ahmad, Mohamed Hefnawy, Mohammed Al-Yahya, Omer A. Basoudan, and Syed Rafatullah / Mediators of Inflammation, Volume 2014 (2014) /
http://dx.doi.org/10.1155/2014/983952
(25) Evaluation of beetroot (Beta vulgaris L.) leaves during its developmental stages: a chemical composition study / Polyana Batoqui França Biondo; Joana Schuelter Boeing; Érica Oliveira Barizão; Nilson Evelazio de Souza; Makoto Matsushita; Claudio Celestino de Oliveira; Marcela Boroski; Jesuí Vergílio Visentainer* / Food Sci. Technol (Campinas) vol.34 no.1 Campinas Jan./Mar. 2014 Epub Mar 11, 2014 / http://dx.doi.org/10.1590/S0101-20612014005000007
(26) Use of Beta vulgaris as natural coloring agent for foods and cosmetics in Libya / Marwa M. Dlim , Sami G. Alsabri , Sofian S. Mohamed , Abdulmottaleb E. Zetrini / Journal of Chemical and Pharmaceutical Research, 2013, 5(11): pp 340-345 / ISSN: 0975-7384
(27) ANTIMICROBIAL ACTIVITY OF ETHANOLIC ROOT EXTRACT OF BETA VULGARIS / Krishnaraj R*, Murugan R, Premlal KR / Journal of Science, Vol 6, Issue 1, 2016, pp 39-40
(28) Effects of Chard (Beta vulgaris L. var. cicla) Extract on Oxidative Injury in the Aorta and Heart of Streptozotocin-Diabetic Rats / G. Sener, Ö. Saçan, R. Yanardağ, and G. Ayanoğlu-Dülger. / Journal of Medicinal Food. July 2004, 5(1): pp 37-42. / doi:10.1089/109662002753723205.
(29) Prevention of High Blood Pressure by the Beeta Vulgaris Root Solution and Allium Sativum Bulbs Solution by the Survey Study / Lavanya Rentikota / Health Science Journal
(30) Beta vulgaris / Wikipedia
(31) The antioxidant activity of Beta vulgaris leaf extract in improving scopolamine-induced spatial memory disorders in rats / Shadieh Hajihosseini, Mahbubeh Setorki, Zahra Hooshmandi / Vicenna Journal of Phytomedicine, Sept-Oct 2017; 7(5): pp 417-425 / PMID: 29062803
(32) Effect of Beta vulgaris Extract on Liver Enzymes in Patients with Non-Alcoholic Fatty Liver Disease: A Randomized Clinical Trial / Nikta Afzali, Seyed Shayan Ebadi, Hasan Afzali, Habibollah Rhamini et al / Hepatitis MonthlyL International Monthly Journal in the Field of Hepatology
(33) Antibacterial activity: Amarathus graecizans; Beta vulgaris; Polyconum patulum; Rumex obtusifolius: Ethanolic extract / Haniyeh Koochak, Seyyed Mansour Seyyednejad, Hussein Motamedi / Asian Paciific Journal of Tropical Medicine, March 2010; 3(3): pp 180-184 / DOI: https://doi.org/10.1016/S1995-7645(10)60004-1
(34) Studies on evaluation of physical and chemical composition of beetroot (Beta vulgaris L.) / Kale RG, Sawate A R, Kshirsagar R B, Patil B M, and Mane R P / International Journal of Chemical Studies, 2018; 6(2): pp 2977-2979 / pISSN: 2349-8528 eISSN: 2321-4902
(35) Phytochemical study of the Iraqi Beta vulgaris leaves and itw clinical applications for the treatment of different dermatological diseases / Nidhal K Maraie, Thukaa Z Abdul-Jalil, Anas T Alhamdany, and Hassan A Janabi / World Journal of Pharmacy and Pharmaceutical Sciences, 2014; 3(8): pp 5-19 / ISSN: 2278-4357
(36) Chemical Composition and Biological Studies of the Essential Oil from Aerial Parts of Beta vulgaris subsp. maritima (L.) Arcang. Growing in Tunisia / Afifa Zardi-Bergaoui et al / Chemistry & Biodiversity, Oct 2017; 14(10): e1700234 / DOI: https://doi.org/10.1002/cbdv.201700234
(37) Improvement of hypertension, endothelial function and systemic inflammation following short-term supplementation with red beet (Beta vulgaris L.) juice: a randomized crossover pilot study / S Asgaru, M R Afshani, N Sarrafzadegan et al / Journal of Human Hyperytension, 2016; 30: pp 627-632 /
DOI: https://doi.org/10.1038/jhh.2016.34
(38) Antioxidant and antiacetylcholinesterase activities of chard (Beta vulgaris L. var. cicla) / Ozlem Sacan, Refiye Yanardag / Food and Chemical Toxicology, May 2010; 48(5): pp 1275-1280 /
DOI: https://doi.org/10.1016/j.fct.2010.02.022
(39) Evaluation of pigments from methanol extract of Tagetes erecta and Beta vulgaris as antioxidant and antibacterial agent / Mariya Sanni, Reena Lawrence, and Kapil Lawrence / Natural Products Research, 2018; 32(10) / DOI: https://doiorg/10.1080/14786419.2017.1326037
(40) Antitumor and Antiviral Activity of Columbian Medicinal Plant Extracts / L A Betancur-Galvis, J Saez, H Granados, A Salazar, J E Ossa / Memorias do Instituto Oswaldo Cruz, July 1999; 94(4): pp 531-535 /
DOI: https://doi.org/10..1590/S0074-02761999000400019
(41) Effect of red beetroot (Beta vulgaris L.) intake on the level of some hematological tests in a group of female volunteers / Nora M Al-aboud / ISABB Journal of Food and Agriculture Science, Feb 2018; 8(2): pp 10-17 / ISSN: 1937-3244 / DOI: 10.5897/ISABB-JFAS2017.0070
(42) Effect of Beta vulgaris L. on cholesterol rich diet0induced hypercholesterolemia in rats / Mohammed Al-Dosari, Saleh Alqasoumi, Majid Ahmad, Mohammed Al-Yahya, M Nazam Ansari =, Syed Rafatullah / Farmacia, 2011; 59(5) pp 559-679
(43) Biochemical and molecular studies on the possible influence of the Brassica oleracea and Beta vulgaris extracts to mitigate the effect of food preservatives and food chemical coloorants on albino rats / Mohammed A A Sarhan, Ali A Shati. fahmy G Elsaid / Saudi Journal of Biological Sciences, Sept 2014; 21(4): pp 342-354 / DOI: https://doi.org/10.1016/j.sjbs.2013.11.004
(44) Medicinal Foodstuffs. III. Sugar Beet (1): Hypoglycemic Oleanolic Acid Oligoglycosides, Betavulgarosides I, II, III, and IV from the Roots of Beta vulgaris (Chenopodiaceae) / Masuki Yoshikawa, Toshiyuki Murakami, Nobutoshi Murakami et al / Chemical and Pharmaceutical Bulletin. 1966; 44(6): pp 1212-1217 / DOI: https://doi.org/10.1248/cpb.44.1212
(45) Study of medicinal plants in Aravali regions of Rajasthan for treatment of kidney stoone and urinary tract troubles / Neha Sharma, Babeet Singh Tanwer and Rekha Vijayvergia / International Journal of PharmTech Research, Jan-Mar 2011; 3(1): pp 110-113 / ISSN: 0974-4304
(46) Nutritional and functional potential of Beta vulgars cicla and rubra / Paolino Ninfali, Donato Angelino et al / Fitoterapia, Sept 2013; Col 89: pp 188-199 / DOI: https://doi.org/10.1016/j.fitote.2013.06.004
(47) Effect of Beta vulgaris Linn. Leaves Extract on Anxiety- and Depressive-like Behavior and Odixative Stress in Mice After Acute Restraint Stress / Kunjbihari Sulakhiya, Vikas Kumar Patel, Rahul Saxena, Jagrati Dashore, Amit Kumar Srivastava and Manoj Rathore / Pharmacognosy Research, Jan-Mar 2016; 8(1): pp 1-7 / PMID: 26941529 / DOI: https://doi.org/10.4103/0974-8490.171100
(48) Ameliorative Effect of Beta vulgaris Root Extract on Chlorpyrifos-Induced Oxidative Stress, Infammation and Liver Injury in Rats / Gadah Albasher, Rafa Ameer, Ayman M Mahmoud et / Biomolecules, 9(7) / DOI: 10.3390/biom9070261
(49) Triterpene Saponin Content in the Roots of Red Beet (Beta vulgaris L.) Cultivars / Agnieszka Meoxzek, Ireneusz Kapusta, Bogdan Janda,, Wirginia Janiszowska / Journal of Agricultural and Food Chemistry, 2012; 60(50): pp 12397-12402
(50) Kinetic Properties of Peroxidase Enzymes from Chard (Beta vulgaris Subspecies cicla) Leaves / Ercan Bursal / International Journal of Food Properties, 2014; 16(6) / DOI: https://doi.org/10.1080/10942912.2011.585729
(51) Synergistic protective effect of Beta vulgaris with meso-2,3-dimercaptosuccinic acid against lead-induced neurotoxicity inn male rats / Nadia Z Shaban, Sara E Abd E;-Lader, Noha H Habashy et al / Scientific Reports, 2021; 11, Art No: 252 / DOI: https://doi.org/10.1038/s41598-020-80669-4
(52) Prophylactic Impact of Beta vulgaris L in Ameliorating Cyclosporine A-Induced Hepatotoxicity in Rats / Wafa I Albalawi, Nadin A Abdul Majid, Iman A Sharaf / International Journal of Pharmaceutical Research & Alied Sciences, 2019; 8(2): pp 22-34 / ISSN: 2277-3657
(53) Sugar Beet Extract (Beta vulgaris L.) as a New Natural Emulsifier: Emulsion Formulation / Theo Ralla, Hanna Salminen, Jochen Weiss et al / Journal of Agricultural and Food Chemistry, 2017; 65(20): pp 4153-4160 DOI: https://doi.org/10.1021/acs.jafc.7b00441
(54) Beetroot (Beta vulgaris) rescues mice from y-ray irradiation by accelerating hematopoiesis and curtainling immunosuppression / Jinhee Cho, So Jin Biing, Areum Kim, Nam Ho Lee, Sang-Hee Byeon / Pharmaceutical Biology, 2017l 55(1) / DOI: https://doi.org/10.1080/13880209.2016.1237976
(55) Aqueous Fraction of Beta vulgaris Ameliorates Hypoglycemia in Diabetic mice due to Enhanced Glucose Stimulated Insulin Secretion, Mediated by Acetylcholine and GLP-1, and Elevated Glucose Uptake via Increased Membrane Bound GLUT4 Transporters / Ashraf Ul Kabiir, Mehdi Bin Samad, J M A Hannan et
al / PLOS ONE, Feb 2015 / DOI: https://doi.org/10.1371/journal.pone.0116546
(56) Toxicity Study of Beetroot (Beeta vulgaris) Extract in Normal Sprague Dawley Rats / Fidelis E Olumese, Henrietta A Oboh / NISEB Journal, 2017; 17(3)
(57) Neuroprotective potential of Beta vulgaris L. in Parkinson’s disease / Vandana S Nade, Laxman A Kawale, Shankar S Zambre, Amit B Kapure / IIndian Journal of Pharmacology, Jul-Aug 2015; 47(4): pp 403-408 / PMID: 26288473 / DOI: 10.4103/0253-7613.161263
(58) The Ethanolic Extract of Beetroot (Beta vulgaris) Ameliorates Some Red Cell Parameters in Phenylhydrazine-Induced Anaemic Rats / Favour, Nyoh Beshel, Justin Atiang Beshel and Ekamma Edet Ante / IOSR Journal of Nursing and Health Science, Jul-Aug 2018; 7(4) Ver X: pp 27-30 /
DOI: 10.9790/1959-0704102730
(59) Beet root (Beta vulgaris) protects lipopolysaccharide and alcohol-induced liver damage in rat / Bae-Hwan Kim, Se-Hoon Jung, Suryun Jung / Toxicological Research, 2020; 36: pp 275-282 /
DOI: https://doi.org/10.1007/s43188-019-00030-4
(60) Phytosynthesis and Characterization of Copper Oxide Nanoparticles using the Aqueous Extract of Beta vulgaris L. and Evaluation of their Antibacterial and Anticancer Activities / Rajkuberan Chandrasekaran, Sangilimuthu Alagar Yadav and Sivaramakrishnan Sivaperumal / Journal of Cluster Science, 2020; 31: pp 221-230 / DOI: https://doi.org/10.1007/s10876-019-01640-6
