
Notes on Alginate
Introduction
“Increasing the value of under-utilized muscles of food animals has been the target of many published reports involving alginate. Many of these muscles, usually originating from the hind leg or shoulder area, are underutilized due to lower palatability. Connective tissue removal, tenderization, and comminution improve the raw material. Restructuring allows for the recombination of the meat into a shape that is easily recognized and handled. Early reports on restructuring meat products promote the use of salt and/or phosphate to aid in the extraction of proteins which hold the meat particles together once the product is heat processed.
However, in the raw state, a product made in this way must be frozen in order to retain its integrity. Marketing an unfrozen or uncooked restructured product processed using only salt and phosphate is virtually impossible. Storage stability and visual appearance of these products are also a concern since products containing salt have a tendency to oxidize at a faster rate.” (Tarté, 2009)
To overcome the need to freeze or cook restructured products, a binder was sought that becomes active without heat and holds the pieces of meat together in both the raw and cooked state. Sodium alginate, a common ingredient in the food industry, is a hydrocolloid that gels in the presence of multivalent ions and forms a heat-stable gel. Utilizing this process has the added advantage of not requiring the inclusion of salt or phosphate for protein extraction and, because of the heat-stable gel formed, the integrity of the structured product withstands normal cooking temperatures for meat products (Clarke, Sofos, & Schmidt, 1988; Means, Clarke, Sofos, & Schmidt, 1987; Means & Schmidt, 1986)
Description and Origin
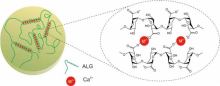
“Alginate is an anionic polymer that occurs naturally in brown algae (Phaeophyceae), normally present on the cell walls of these organisms.” (Pereira, 2019)
“Alginate is a structural element designated to be the seaweed’s main skeletal compound likewise the cellulose function in terrestrial plants, with the gel located in the cell walls and intercellular matrix conferring the mechanical strength and flexibility necessary to withstand the force of the water in which the seaweed grows. Moreover, this function is reflected in the compositional difference of alginates in different seaweeds.” (Pereira, 2019)
“Alginate varies in composition of the algae from 20 to 60% dry matter, but on average brown algae species has 40% alginate. Alginate in brown algae occurs as gels containing sodium, calcium, strontium, magnesium, and barium ions.” (Pereira, 2019)
“Alginate is not a compound exclusively of brown algae because there are bacteria that can also produce alginate, but currently all commercial alginate is extracted from algae biomass.” (Pereira, 2019)
“Alginic acid is a copolymer of two uronic acids, mannuronic and guluronic, is extracted from brown seaweed, and has the unique property of forming a gel in the presence of a divalent cation. When producing alginates, the uronic acids are converted into their salt forms, mannuronate (M) and guluronate (G) through a neutralization step. In the natural state, M and G units can be linked together in one of three blocks: MM, GG, or MG (GM). The proportion, distribution, and length of these blocks determine the chemical and physical properties of the molecules. The reactivity with calcium and the consequent gelling capacity is a direct function of the average length of the G blocks.” (Tarté, 2009)
“Alginates containing high levels of GG fractions possess the ability to form strong gels. These fractions are more abundant in the stems of the seaweed, while the leaves contain alginic acids with higher proportions of M fractions.” (Tarté, 2009)
Discovery
“Alginic acid was first discovered and patented (patent date: 12 January 1881) by the British chemical scientist E. C. C. Stanford, and he continued the work on its discovery, contributing to the elucidation of the chemical structure of alginic acid [3]. The Stanford patent explains how the alginate can be extracted by soaking the algae with water or diluted acid, then extracting with sodium carbonate, and then precipitating the alginate present in the solution by addition of acid.” (Pereira, 2019)
“In the second decade of the twentieth century, some scientific groups working separately with alginate found that uronic acid was one of the constituents of alginic acid. Moreover, this discovery led to further study in the years to come. These investigations led to the discovery of D-mannuronic acid in hydrolyzed alginate samples. The nature of the bonds in the uronic acid residues in the alginate was identical to that in the cellulose, through the β 1, 4 bond.” (Pereira, 2019)
“It was only in the 1950s that with the work of Fischer and Dörfel, through a chromatographic study of uronic acids, the presence of a different uronic acid from what had been identified was discovered, identifying this new acid with L-guluronic acid. And that acid had a considerable quantity in the sample analyzed, and as such, a quantitative method was developed to determine the two acids present in alginate, mannuronic acid and guluronic acid.” (Pereira, 2019)
“Thereafter, alginate was identified as a binary copolymer composed of residues of guluronic and mannuronic acids, but in general, it was reported that alginic acid was chemically homogeneous and of equal chemical structure, independent of the raw material from which it was extracted.” (Pereira, 2019)
“This principle had to be scientifically proven, and the alginate had to be fractionated chemically from different sources to prove the theory. The development of fractionation techniques was done mainly by Haug, who helped the characterization alginate as a block copolymer and in the correlation between the block structure and the physical properties of an alginate with that composition.” (Pereira, 2019)
Dennis McHugh did a great overview of its discovery and writes, “Alginic acid was first discovered by Stanford (1881). An excellent history of the evolution of the alginate industry has been written by Booth (1975). He traces a path from Stanford’s successful exploitation of crude extracts to the failure by F.C. Thornley, in Orkney about 1923, to establish a briquette business based on using alginate as a binder for anthracite dust. Thornley moved to San Diego and by 1927 his company was producing alginate for use in sealing cans. After some difficulties the company changed its name to Kelp Products Corp. and in 1929 it was reorganized as Kelco Company. Production in the United Kingdom was established in the period 1934-1939 and in Norway after World War II. It is estimated that there are 17 factories in 9 different countries (ITC, 1981), excluding the People’s Republic of China. The two largest producers, Kelco Company in USA and Alginate Industries Ltd in UK, have been acquired by Merck and Co. Inc., USA; these combined companies produce about 70% of the world’s alginate. The next largest producer is Protan A/S of Norway, followed by companies in Japan and France (ITC, 1981). Production in China is increasing and is now 7 000-8 000 tonnes per annum.”
“Some of the early patents still provide useful basic information about alginate extraction (Thornley and Walsh, 1931; dark and Green, 1936; Green, 1936; Le Gloahec and Herter, 1938; Le Gloahec, 1939) as does work published by the former Institutes of Seaweed Research in Scotland and Norway (Black and Woodward, 1954; Haug, 1964) and more recently by Braud, Debroise, and Pérez (1977). Processes used in Japan have been described by Okazaki (1971).
The minimal requirements for the profitable operation of an alginate extraction plant have been estimated by Moss and Doty (1987). They discuss the minimal seaweed input, colloid output and capital investment needed; they also list estimates of production costs. This analysis is made for agar and carrageenan as well as alginate.” (McHugh)
Functional Characteristics and Properties
“Alginates are soluble in cold water and therefore do not require heating to form gels. These gels develop upon exposure to multivalent cations. Because of its commonality and safety, calcium is usually the cation of choice.” (Tarté, 2009) Other cations often used in the formation of a matrix of alginic acid with bound cations are sodium and magnesium. (Pereira, 2019)

“The solubility of the calcium source is one of the criteria used to control the rate of gel formation. A calcium salt solution will cause alginate in solution to gel on contact. Continuous exposure of the alginate to the calcium solution will increase the firmness of the gel, as more calcium diffuses into the gel and binds to the G blocks within the alginate structure. This process, used for encapsulating liquids, can be used for forming a gel layer, or skin, on the surface of meat products. The addition of alginate to a meat blend followed by bathing or showering in a calcium chloride solution results in the formation of skin on the surface. Extended exposure time to the calcium chloride solution will increase the thickness of the skin and, if long enough, will eventually create a gelled structure throughout the product due to calcium diffusivity. Time of complete gelling will be dependent on the thickness of the structure. This process of forming an alginate gel is referred to as external gelling.” (Tarté, 2009)
“For internal gelling, sodium alginate and the calcium source are added directly to the meat mixture and allowed to form a gel over time. The success of this process is based on ensuring the complete hydration of alginate and the proper timing of calcium release, which can be controlled by the solubility of the calcium source or by the addition of a sequestrant. A gel formed too quickly has the potential to break apart during the mixing or stuffing process. Because calcium alginate gels do not re-heal, if the gel is broken during the structuring process, it could appear that it did not form in the first place. Calcium chloride would not be the calcium source of choice for this type of application. More commonly, a less soluble calcium source is used. The process for making structured meat using alginate gels was patented by researchers at Colorado State University (Schmidt & Means, 1986).” (Tarté, 2009)
Alginate, Calcium Carbonate and GDL
“The patent describes a method to form structured meat products using sodium alginate, calcium carbonate, and glucono-delta-lactone (GdL). Sodium alginate, once added to the meat block, is hydrated by the moisture in the meat. Calcium carbonate has low solubility and gives the processor time to stuff the meat mixture into casings. GdL decreases pH over time, which causes the solubility of calcium carbonate to increase, thus releasing calcium and resulting in the formation of an alginate gel able to entrap the meat particles. Once the gel has set, the product is sliced into steaks or chops. Since the calcium alginate gel is heat stable, the product will not fall apart during cooking.” (Tarté, 2009)
“To assure optimum gelling properties, it is essential that the alginate be fully hydrated and evenly distributed in the mixture. Adding alginate directly to the meat source allows the alginate to hydrate by drawing moisture from the meat. To speed up gel formation, sodium alginate should be hydrated with water prior to addition to the meat mixture. The action of a sequestrant is to chelate calcium, with the objective being to slow down the gelation mechanism of sodium alginate. There is evidence that slowing gelation can also increase gel strength, most likely due to a more ordered formation of the gel structure.” (Tarté, 2009)
“Conditions that retard intermolecular interactions will result in a more homogeneous and regular network and consequently a stronger gel (Bernal et al., 1987). The sequestrant most often used is phosphate. The effectiveness of the sequestrant is influenced by solubility. For example, at equal phosphate levels, sodium tripolyphosphate is more effective than tetrasodium pyrophosphate at retarding gel formation.” (Tarté, 2009)
Texture – Structured Pork Rolls
The texture of structured meat is a primary focus among meat processors. Structured meat should be superior to ground meat, without having a gel-like texture. The mechanical properties of structured meat are influenced by size and shape of the raw material (Berry & Civille, 1986) as well as sodium alginate/calcium ratio (Trout, Chen, & Dale, 1990). Devatkal and Mendiratta (2001) found the mechanical properties of structured pork rolls were superior for salt-phosphate rolls in the cooked state but were better for alginate/lactate gels in the raw state.” (Tarté, 2009)
Alginate Structured Meat with Added Water
“Raharjo et al. (1995) found bind force to be higher for calcium alginate-structured meat made with sliced meat and added water than for a no-additive control.” (Tarté, 2009)
“As Schaake, Means, Moody, Boyle, and Aaron (1993) found, calcium alginate steaks had significantly lower bind force compared to salt and phosphate restructured steaks. Interestingly, however, sensory panels rated calcium alginate steaks equivalent to those made with salt and phosphate, the only exception being sliced meat with added water (Raharjo et al., 1995) being lower in sensory ratings.” (Tarté, 2009)
Dosage
“Tensile strength of structured products is a function of both alginate and calcium. In a study by Trout (1989), cooked tensile strength of restructured pork chops was measured on factorial arrangements of calcium and alginate concentrations. Sodium alginate alone decreased tensile strength, as it interferes with protein interactions. A slight increase in tensile strength was observed when 0.13% calcium was added to 0.7% alginate. There was no increase in tensile strength with further increases in alginate. However, an increase in calcium at both levels of alginate tested (0.7% and 1.4%) resulted in an increase in tensile strength. Means and Schmidt (1986) indicated the ideal alginate : calcium ratio was 2.5 g alginate:0.18 g calcium ion. Ensor, Ernst, Sofos, and Schmidt (1986) made acceptable structured turkey meat using 0.4–1.0% sodium alginate, 0.075–0.1875% calcium carbonate, and 0.6% lactate. Later, Ensor, Sofos, and Schmidt (1990) found 0.4% sodium alginate, 0.075% calcium carbonate, and 0.6% lactate were optimal for use in structured meat products. Basic differences in optimal use level will result with different raw materials. As the divalent ion concentration increases, there is an increase in the strength of the alginate gels, resulting from multiple molecular interactions. Along with the increase in gel strength, however, increases in calcium level can also result in an increase in syneresis or purge, as stronger gels will tend to push more water out of the structure. Decreasing the amount of calcium added to the formula will lower the gel strength and reduce the associated syneresis. (Tarté, 2009)
“The gelling mechanism for alginate is well known. However, the extent to which the gelling mechanism is influenced by the presence of muscle proteins has not been elucidated. A few studies have been conducted to determine if proteins are part of the gelling mechanism in meat applications. Imeson, Ledward, and Mitchell (1977) observed considerable changes in UV–visible absorption of myoglobin and serum albumin caused by alginate and certain other polysaccharides. These absorption spectra indicated some type of interaction between alginates and proteins, which the authors attributed to electrostatic forces. Bernal et al. (1987) later confirmed the involvement of electrostatic forces with crude myofibrillar protein.” (Tarté, 2009)
“However, Xiong and Blanchard (1993) showed no change in absorption spectra when scanning various combinations of salt-soluble proteins (SSP) and alginate but did find a reduction in the gelling suspension. Offering an explanation for this unpredicted result, the authors surmised that there may be minimal hydrocolloid–protein interactions in the initial phase of SSP gelation (structural unfolding) but, as the gelling point is approached, alginate interferes with the gel network formation. It could also be that alginate has no influence on hydrophobic groups within the protein molecule but has some influence on electrostatic bonding. This influence of alginate could also be observed in the final gelled product as lower gel firmness. Schaake et al. (1993) reported that dried egg albumin did not enhance the performance of the alginate binding system in structured beef steaks, while Ensor, Sofos, and Schmidt (1991) suggested a change in the physical state of proteins in a structured meat system with the addition of algin/calcium as evidenced by differential scanning calorimetry (DSC). Mei, Chan, and Lin (2002) indicated that alginate appeared to protect the myosin heavy chain through heat denaturation, suggesting that the alginate surrounds and protects the protein, and may, consequently, interfere with the protein gelling mechanism. This was also seen in surimi, where alginate provided protection against frozen storage. The interaction between protein and alginate has yet to be completely defined. However, in most cases, higher levels of protein will interfere with the alginate gelling mechanism. Meat products using alginate gel technology should be treated like ground meat from a food safety standpoint (Ortega-Valenzuela, Phebus, & Thippareddi, 2001) and, therefore, should be cooked to internal temperatures that ensure product safety.” (Tarté, 2009)
Limiting Factors
“This could limit their acceptance as replacements for intact muscle. Making products using batch-type systems is another limitation for the acceptance of alginate gel technology. There are no known methods to process structured meat products on a continuous basis. Wotherspoon (1988) was granted a patent teaching the continuous production of reconstituted pet food chunks from comminuted materials. Modifications of this procedure may be used to make raw materials for further processed meat products. Besides increasing the value of under-utilized muscles, meat processors can take advantage of alginate characteristics in other ways.” (Tarté, 2009)
Alginate and MDM
“Mechanically separated muscle has limited use due to its contribution to a reduction in firmness of the final product. Using alginates to structure these products improves the texture of the final product, with the added advantage of improving cook yield (Lamkey, 2006).
On addition of sodium alginate to a calcium solution, interfacial polymerization is instantaneous with precipitation of calcium alginate followed by a more gradual gelation of the interior as calcium ions permeate through the alginate. Entrapment in insoluble calcium alginate gel is recognized as a rapid, non-toxic, inexpensive, and versatile method for immobilization of enzymes and cells (Entrapment in Calcium Alginate; Jane E. Fraser, Gordon F. Bickerstaff )
Suggested dosage: 6g Alginate per kg MDM and water (Water = 30% MDM weight). Mix for 20 mins.
Alginate in a Protein Emulsion
“Sodium alginate can be used to structure mixtures of protein and water, where the protein source can range from 25% to 70% of the formulation. The boundaries are limited only by the texture desired. Reducing the amount of protein creates a firm, gel-like texture while increasing the protein results in softer but more meat-like texture.” (Tarté, 2009)
Alginate in Sausage Production
If used in a fine emulsion sausage or polony formulation, it has the effect of increasing firmness and prevents jelly and fat cookout. It generally stabilises the mixture. A mixture is suggested to contain:
- animal protein
- e401 sodium alginate
- e516 calcium sulphate or calcium carbonate
- e4450 diphosphate or use Trisodium phosphate which slows down the reaction. The calcium sulphite reacts with the trisodium phosphate to form calcium phosphate preventing the calcium sulphate reacting with the alginate to form a gel. This will happen until the trisodium phosphate is used up and then the gelling reaction will take place.
- maltodextrin
- e330 citric acid
- e150c ammonia caramel (colour)
- flavouring substance
- vegetable oil
- e414 Arabic gum (thickening agent)
- e307 alpha-tocopherol (Vitamin E)
- e120 cochineal (colour) (with e524 sodium hydroxide)
This same mix is sold under the trade name, Prallo.
As Fat Replacer
Lin and Keeton (1998) suggested a combination of sodium alginate and carrageenan can replace fat in precooked beef patties with similar textural properties. In this case, alginate is used in its non-gelled form. Means and Schmidt (1986), however, found off-flavours associated with higher alginate levels and attributed it to non-gelled alginate. It is, therefore, recommended that sensory attributes on non-gelled alginate products be evaluated.” (Tarté, 2009)
A fat replacer is being marketed, also under the trade name, Prallo, containing
- E401 sodium alginate
- E516 calcium sulphate
- E450 diphosphates
- soya fibre
- bovine collagen
- E1442 modified starch
As a Meat Coating System – Meat Patty Application
“Alginate, like most carbohydrates, can also form films used to protect the surface of fresh meat products. A patent by Earle and McKee (1976) describes the use of alginate in combination with starch to form a coating for the protection of fresh beef carcasses. Williams, Oblinger, and West (1978) concluded that a calcium alginate coating significantly decreased shrink in coated vs. uncoated steaks and also helped maintain the oxymyoglobin colour for a longer period of time. Warmed-over flavour was eliminated in precooked, alginate-coated patties as judged by sensory scores and TBA values (Wanstedt, Siedeman, Donnelly, & Quenzer, 1981). Patties having a calcium alginate coating was juicier and more desirable in texture and overall palatability than raw or precooked patties without calcium alginate. A reduction in oxidation, as well as an improvement in moisture retention, were shown to be advantages of having alginate in a presoak for rabbit muscle that was irradiated with 5 KGy.” (Tarté, 2009)
Agricultural applications
The agricultural application of Alginate is fascinating and provides clues to its value in meat processing. “Alginate present in the brown algae (in the form of alginic acid) constituted a functional element of the traditional fertilizers, allowing the water retention in the soils. So, the principal function of alginate on agricultural area was as a soil conditioner. Being a superabsorbent (SAP) or water-retaining material is an advantage of alginate. They are natural materials that can absorb large amounts of water, as much as hundreds of times their own mass. These alginates are generally known in agriculture as nonionic or ionic moisture-holding hydrogels for increasing soil water retention, which is a basic soil property.” (Tarté, 2009)
“The reservation of moisture or water in the soil is the major process consequence in which all plantations depend. The large pore spaces in arenaceous soils restrain the soil from holding water, and the soil dries out regularly, and precious nutrients wash away past the plant roots. The inclusion of alginate can solve the lack of retention of water and raise nutrient disponibility. High-capacity absorbents definitely can upsurge the water-retention capacity in such soils.” (Tarté, 2009)
“Superabsorbents (SAP) in agricultural areas have been designed and developed to provoke an enrichment of the abiotic properties of soil by rising their water-retention ability, developing a better water usage efficiency, enhancing soil permeability and infiltration rates, contributing to lower the irrigation frequency, lowering the compaction shift, preventing erosion and water drainage, enhancing plant performance, increasing soil aeration, lowering the dissolution of fertilizers, developing a better adsorption capacity or enhancing the uptake of some nutrient elements by the plants, and provoking a raise of the microbial activity.” (Tarté, 2009)
“The alginate of seaweed directly suppresses the pathogens. Indeed, alginate pellets developed as carrier material for biocontrol agents have been reported to reduce multiplication of Rhizoctonia (fungi) disease in potato, while incorporation of Ascophyllum nodosum extract into the planting medium caused delay and reduced incidence of Verticillium (fungi) wilt of pepper plants. Therefore, it’s proven that alginates are involved in host defense mechanisms. Of particular interest in agriculture are those that elicit defensive responses resulting in protection against pathogens or insect damage.” (Tarté, 2009)
“In other cases, the alginate will have other particular function, as the main characteristic of alginate as product principal emulsifier and to delivery control of actives ingredients in agricultural field. The active ingredient is mixed with alginates for their safer, easier, and more accurate handling as well as for their effective application in the field and, at the same time, preventing the immediate release of the active ingredient, so the main drawback associated with these formulations can be avoided. These alginate-based systems are able to deliver the active ingredient gradually for a long period of time in a specified target with a desired rate. The controlled-release systems do not release the active ingredient at once; this technique therefore lowers the pesticide residues in soil and thus reduces the direct effect of pesticide. After their degradation, these are helpful as compost in the field.” (Tarté, 2009)
“Alginate is also used as an inoculant carrier for plant growth-promoting bacteria and for bacteria with biodegradation ability.” (Tarté, 2009)
Food applications
“Alginates are commonly used in the food industry as natural additives; they have codes from the European Union as food additives, and these codes vary with the ion type associated with alginic acid.” (Tarté, 2009)
“European codes for alginates are as follows: alginic acid, E400; sodium alginate, E401; potassium alginate, E402; ammonium alginate, E403; calcium alginate, E404; and propylene glycol alginate, E405.” (Tarté, 2009)
“Seaweed polysaccharide applications in food industries are based mainly on their stabilizing, emulsifying, and gel-forming ability. They are widely used as food additives in jams, jellies, ice creams, dairy products, etc., to improve and stabilize the structure of food.” (Tarté, 2009)
“Water-in-oil emulsions likewise the mayonnaise and the salad fillings are less liable to fractionate toward their original oil and water phases if thickened with alginate. When the emulsion is acidic, the sodium alginate will precipitate into insoluble alginic acid forms; to resolve this problem, propylene glycol alginate (PGA) is used for acidic emulsions, because this compound is stable in mild acid conditions.” (Tarté, 2009)
“The advantage of alginate in the food industry is that humans do not have enzymes to break the molecule; therefore, alginate behaves as a dietary fiber, enhancing the satiety and reducing the food intake of humans, lowering the energy intake by human, and preventing obesity.” (Tarté, 2009)
Application Notes
Sodium algiante is generally used in meat systems at 0.5-2%.
1. Dissolve sodium alginate into the water at an 8% solution, stir with a high-speed blender to be paste (within 3 mins ); or dissolve sodium alginate into appropriate vegetable oil (1:1-2), stir it to a paste, then pour it to create dogfood.
2. Moulding meat to different shapes, leave it 6-10h at 4 ℃ and thereafter transfer it for cold storage and slice.
Regulatory Status
“Based on current USDA regulations, a mixture of sodium alginate (not to exceed 1% of product formulation), calcium carbonate (not to exceed 0.2%), and calcium lactate/lactic acid (or GdL) (not to exceed 0.3%) is permitted for use in restructured meat food products to bind meat pieces. The entire mixture is not to exceed 1.5% of product at formulation and it must be added dry (CFR, 2007b). For ground and formed raw or cooked poultry pieces, sodium alginate cannot exceed 0.8%, calcium carbonate cannot exceed 0.15%, and calcium lactate/lactic acid cannot exceed 0.6%. The entire mixture cannot exceed 1.55% of product formulation and it also must be added dry (CFR, 2007b). The USDA also allows for the application of an alginate film on freshly dressed meat carcasses to reduce cooler shrinkage and help protect surface (CFR, 2007b). A mixture containing water, sodium alginate, calcium chloride, carboxymethylcel-lulose, and corn syrup solids may not exceed 1.5% of hot carcass weight when applied and chilled weight cannot exceed hot carcass weight (no added water). In the European Union, sodium alginate is listed in the group of approved emulsifiers, thickeners, stabilizers, and gelling agents for use “quantum satis,” and has been assigned the reference number E 401 (European Parliament and Council, 2006) .” (Tarté, 2009)
Oake Woods & Co Notes
Sodium alginate is a commonly used gelling agent that can form a gel when combined with calcium ions. The process of gel formation occurs when the calcium ions cross-link the chains of sodium alginate, creating a 3D network that solidifies into a gel.
The concentration of calcium ions required for gel formation depends on the concentration of sodium alginate present in the solution. Specifically, a lower concentration of sodium alginate will require a higher concentration of calcium ions for gel formation, because there are fewer chains to be cross-linked. Conversely, a higher concentration of sodium alginate will require a lower concentration of calcium ions, as there are more chains available for cross-linking.
Sodium chloride can potentially interfere with gel formation in sodium alginate, as it can affect the charge interactions between the alginate chains and the calcium ions. Specifically, sodium chloride can screen the electrostatic interactions between the negatively charged carboxyl groups on the alginate chains and the positively charged calcium ions, which can make it more difficult for the chains to cross-link and form a gel.
However, the effect of sodium chloride on gel formation will depend on its concentration in the solution. At low concentrations, sodium chloride may not significantly interfere with gel formation. Conversely, at high concentrations, sodium chloride can completely inhibit gel formation in some cases.
Increasing the calcium content can potentially help to overcome the negative effects of sodium chloride on gel formation, as it can increase the concentration of cross-linking ions available to form the gel. However, there may be limits to how much calcium can compensate for the inhibitory effects of sodium chloride, and there may be other factors to consider, such as the concentration of sodium alginate and the pH of the solution.
Therefore, it may be necessary to do some experimentation to determine the optimal conditions for gel formation in a given system, taking into account the concentration of sodium chloride and calcium ions, as well as other relevant factors.
Both sodium alginate and maltodextrin can be adversely affected by high temperatures during frying. At a temperature of 110°C, both gelling agents may break down or degrade, resulting in a loss of their gelling and water-retaining properties.
However, sodium alginate may be more adversely affected by high temperatures than maltodextrin. This is because the gel formed by sodium alginate is stabilized by ionic cross-links between the alginate chains and calcium ions. High temperatures can cause the breakdown of these cross-links, resulting in a loss of gel structure and water-holding capacity. On the other hand, maltodextrin forms a gel network through hydrogen bonding, which is less sensitive to high temperatures.
Therefore, in general, maltodextrin may be more resistant to high temperatures than sodium alginate during frying. However, it’s important to note that the optimal choice of gelling agent will depend on several factors, such as the specific processing conditions and the desired properties of the final product. It may be necessary to conduct some experimentation to determine the optimal gelling agent and processing conditions for a particular application
Reference
Dennis J. McHugh’ Department of Chemistry, University College, University of New South Wales
Australian Defence Force Academy
Campbell, ACT 2600, Australia
Tarté, R(Ed). 2009. Ingredients in Meat Products Properties, Functionality and Applications. Springer
Pereira, L, and Cotas, J.. 2019. Introductory Chapter: Alginates – A General Overview. Submitted: March 18th, 2019. Reviewed: July 3rd 2019. Published: February 5th, 2020. DOI: 10.5772/intechopen.88381
